Abstract
Examining the association of social determinants of health on critically ill COVID-19 patient outcomes: a retrospective observational study
Christina S. Boncyk MD, MPH, Sonali Shirali MS, Nicholas Flores-Conner MD, Gen Li MStat, MChem, Jacob C. Clifton MS, Pamela Butler BS, Karen Y. McCarthy EdD, Robert E. Freundlich MD, MS, MSCI
Corresponding author: Christina S. Boncyk
Contact Information: Christina.S.Boncyk@vumc.org
DOI: 10.12746/swjm.v13i56.1489
ABSTRACT
Objective: To study the association between air quality index (AQI) and World Health Organization (WHO) Clinical Progression Scale score following Coronavirus disease 2019 (COVID-19) diagnosis.
Methods: Retrospective study of adults admitted to intensive care units between January 1, 2020 and March 31, 2022 with positive SARS-CoV-2 test. We used proportional odds regression to study association between average AQI over 12 months prior to COVID-19 diagnosis and highest WHO Clinical Progression Scale score on 28th day following ICU admission. As sensitivity analyses, we studied average AQI over 2 months prior and 2 weeks prior to COVID-19 diagnosis.
Results: We identified 1,915 patients with median (IQR) AQI of 37 (33–41). Following regression analysis, a 10-unit increase in average AQI values over the past 12 months prior to COVID-19 diagnosis was associated with reduced odds of having a greater WHO Clinical Progression Scale score (OR 0.84; 95% CI 0.72–0.97; p = 0.02). We failed to detect significant association between AQI and WHO Clinical Progression Scale score (p = 0.26 for 2 months AQI, 0.31 for 2 weeks AQI) within sensitivity analyses.
Conclusions: More specific air quality measurements may more accurately identify those at risk for COVID-19 complications following critical illness.
Keywords: Air quality index critical illness, COVID-19, intensive care units, World Health Organization
INTRODUCTION
Patient outcomes are influenced by a multitude of pre-hospital, in-hospital, and patient factors. Importantly, these analyses often exclude social demographic data. The United States Department of Health and Human Services (DHHS), through the Healthy People 2030 framework has identified 5 major groups of Social Determinants of Health (SDOH): (1) Economic stability, (2) education access and quality, (3) health care access and quality, (4) neighborhood and built environment, and (5) social and community context.1 Social determinants of health influence patient access to care, environmental exposures, and societal disadvantages that often interplay to result in poor health outcomes – often independent of race alone.2 While none of these factors are intrinsic to a patient’s identity, many have the potential to impact their intensive care unit (ICU) course4 or recovery after hospitalization.3 In addition, SDOH frequently overlap in the physiologic management of stress and disease pathology that ultimately influence patient outcomes.
Researching SDOH within our patient populations is often difficult and limited by poor documentation or burdensome questionnaires.5 The use of demographic data derived from the electronic health record (EHR) to more accurately represent SDOH holds great promise.2 To better care for our patients and describe factors that influence their outcomes following critical illness, we must further identify and describe the association of SDOH with clinical outcomes. Chronic respiratory illnesses, like asthma, are increasingly prevalent in areas with poor air quality; however, these outcomes within critically ill COVID-19 patients have not been described.6 The connection between air quality and patient outcomes has been studied as it relates to cigarette smoking restrictions and their impact on public health outcomes.7 The Air Quality Index (AQI) has shown great promise as a link between air quality and patient outcomes.
Within the coronavirus disease 2019 (COVID-19) pandemic, literature has found increased virus transmissibility and worsened patient outcomes associated with poor air quality.8 The geographic information system (GIS) provides the tools necessary to incorporate these measures into our research. We used the World Health Organization (WHO) Clinical Progression Scale score as a surrogate for COVID severity9 and evaluate its association with AQI prior to admission among critically ill COVID-19 patients. In addition, we studied the association between AQI and pre-specified secondary in-hospital outcomes. We hypothesized that poor air quality would be associated with higher disease severity for critically ill patients with COVID-19, adding to the impact of inequities in health outcomes.
METHODS
We obtained approval from the Institutional Review Board of Vanderbilt University Medical Center (VUMC) with a waiver for written informed consent due to minimal risk and the retrospective nature of the study that it could not be carried out without the waiver. Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) was used to report this study and the manuscript adheres to the applicable guidelines.10
STUDY POPULATION
We included adult patients (age ≥ 18 years) admitted to a VUMC ICU between January 1, 2020, and March 31, 2022, who had positive test for SARS-CoV-2 performed at VUMC and documented in the EHR. VUMC is a large tertiary referral center for middle Tennessee, southern Kentucky, and northern Alabama. The hospital serves a large and diverse population, and its internal capacity includes six separate adult intensive care units (medical, surgical, neurologic, burn, cardiovascular, and trauma).
DATA COLLECTION
Data were collected through EHR query and included age, sex, home address, date of hospital admission, date of ICU admission, admitting ICU, date(s) of intubation, daily respiratory support, date(s) of extubation, procedures including tracheostomy, date of ICU discharge, date of hospital discharge, date of death, and discharge location, as applicable. We describe the association between AQI and the worst WHO Clinical Progression Scale score on the 28th day following ICU admission. To obtain AQI data, each patient was tied to the air quality monitoring station (https://www.epa.gov/outdoor-air-quality-data/interactive-map-air-quality-monitors) of their respective state and county – using their home address on record. For patients living in an area without a local station monitoring station, the AQI from the 3 closest stations were collected and averaged (as not every state and county combination has a monitoring station).
EXPOSURE AND PRIMARY OUTCOME
Our primary outcome was the WHO Clinical Progression Scale score on the 28th day after ICU admission. This score uses demographic and hospital data to categorize the patient, allowing investigators to track clinically meaningful progression of COVID-19 infected patients.11 There are 4 components to this score: (1) categorization of respiratory support, (2) calculation of SpO2/FiO2, (3) categorization of vasopressors, and (4) classification of end-stage renal disease12 and dialysis. The score is divided into 11 distinct categories of increasing severity: (0) uninfected, (1) asymptomatic, (2) symptomatic; independent, (3) symptomatic; assistance needed (4) hospitalized; no oxygen therapy, (5) hospitalized; oxygen mask or nasal prongs, (6) hospitalized; oxygen by non-invasive ventilation (NIV) or high flow, (7) intubation and mechanical ventilation; pO2/FiO2 ≥ 150 or SpO2/FiO2 ≥ 200, (8) mechanical ventilation or vasopressors pO2/FiO2 < 150 or SpO2/FiO2 < 200, (9) mechanical ventilation and vasopressors, dialysis, or extracorporeal membrane oxygenation (ECMO), and (10) dead (Figure 1).9 Since data could not be collected for patients who were discharged home prior to the 28th day after ICU admission, they were categorized into a single group as either a 0, 1, 2, or 3.
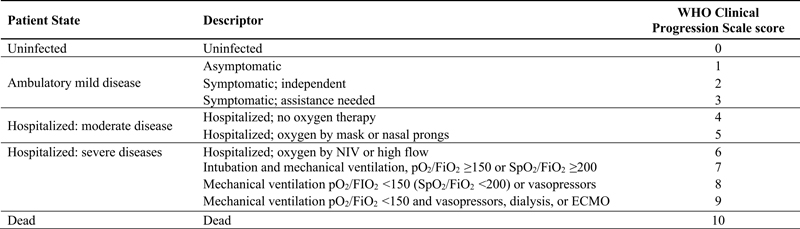
Figure 1. World Health Organization (WHO) Clinical Progression Scale score.
Our intervention of interest was defined as the 12-month average of AQI in the year prior to COVID-19 diagnosis, to accurately describe overall impact of air quality on patients’ respiratory outcomes. The AQI is a numeric metric ranging from 0 to 301 and higher, with larger scores implying worse air quality.
STATISTICAL ANALYSIS
Demographic and procedural variables were summarized with a mean and standard deviation for normally distributed continuous variables, median and interquartile range for non-normally distributed continuous variables and with counts and percentages for categorical variables.
Within our cohort, we used proportional odds regression to elicit the association between AQI and the highest WHO Clinical Progression Scale score on the 28th day following ICU admission. Selected covariates included: age (years) at admission, Elixhauser comorbidity indices (CHF, HTN, chronic pulmonary disease, diabetes, renal failure, liver failure, and metastatic cancer), and body-mass index. Several prespecified secondary outcomes investigated included: ventilator-free days over 28 days following ICU admission, ICU-free days over 28 days following ICU admission, duration of hospitalization, 28-day mortality, and discharge location. We used multinomial logistic regression for discharge location, linear regression for continuous outcomes (ventilator-free days, ICU-free days, and duration of hospitalization), and logistic regression for binary outcome 28-day mortality.
Odds ratios (OR) with 95% confidence intervals (CI) were reported to show the association between the WHO Clinical Progression Scale score and a 10-unit change in average AQI. Mean differences with 95% CI were reported for numeric secondary outcomes ventilator-free days, ICU-free days, and duration of hospitalization. Missing data were handled using multiple imputation. There was no missingness in primary outcome data, and, thus, data were not imputed. Continuous variables were modeled using restricted cubic splines with at least 3 knots. Concordance statistics were reported as a measure of model discrimination. All analyses were performed in SAS 9.4 (SAS Institute Inc., Cary, NC, USA).
SENSITIVITY ANALYSES
As a post hoc sensitivity analysis designed to capture a more comprehensive picture of the impact of AQI on patient outcomes, we further examined the association between the WHO Clinical Progression Scale score with an altered definition of our exposure (average AQI over 12 months prior to COVID-19 diagnosis): (1) the average AQI over 2 months prior to COVID-19 diagnosis and (2) the average AQI over 2 weeks prior to COVID-19 diagnosis.
RESULTS
PATIENT DEMOGRAPHICS
We included 1,915 patients with laboratory-confirmed COVID-19 diagnosis. The average (SD) age was 57.8 (17.8) years, and median (IQR) BMI was 29.7 (25.5–35.2). Median (IQR) AQI scores for each patient in the year prior to COVID-19 diagnosis was 37 (33–41). The most common WHO Clinical Progression Scale score was 1, 2, or 3 – including 1,079 (56.3%) total patients. Detailed patient demographics are displayed in Table 1. Our patients also presented from diverse geographic regions, shown in Supplementary Figure S2.
Table 1. Demographic Characteristics of the Study Sample
| Variables |
Cases (N = 1,915) |
| Age in Years, mean (SD) |
57.8 (17.8) |
| Body Mass Index in kg/m2, median (Interquartile Range) |
29.7 (25.5–35.2) |
| Weight in kg, median (Interquartile Range) |
87.7 (72.8–105.7) |
| Race (%) |
|
| White |
1,423 (74.3%) |
| Black or African American |
314 (16.4%) |
| Other |
178 (9.3%) |
| Gender (%) |
|
| Female |
781 (40.8%) |
| van Walraven Index median (Interquartile Range) |
0 (0–7) |
| Charlson Index median (Interquartile Range) |
1 (0–3) |
| Average AQI in the Year Prior to COVID-19 Diagnosis median (Interquartile Range) |
37 (33–41) |
| WHO Clinical Progression Scale on Day 28 Following ICU Admission (%) |
|
| 1–3 |
1,079 (56.3%) |
| 4 |
51 (2.7%) |
| 5 |
43 (2.3%) |
| 6 |
50 (2.6) |
| 7 |
9 (0.5%) |
| 8 |
69 (3.6%) |
| 9 |
41 (2.1%) |
| 10 |
573 (29.9%) |
| Baseline Elixhauser Comorbidities (%) |
|
| Congestive Heart Failure |
353 (18.4%) |
| Hypertension, Uncomplicated |
826 (43.1%) |
| Hypertension, Complicated |
221 (11.5%) |
| Chronic Pulmonary Disease |
341 (17.8%) |
| Diabetes, Uncomplicated |
369 (19.3%) |
| Diabetes, Complicated |
418 (21.8%) |
| Renal Failure |
382 (20.0%) |
| Liver Disease |
250 (13.1%) |
| Metastatic Cancer |
71 (3.7%) |
| In-hospital Mortality (%) |
576 (30.1%) |
MISSING DATA
There was no missing primary outcome data (i.e., the WHO Clinical Progression Scale score). There were 915 (47.8%) patients without an AQI monitoring station associated with their state and county. For these patients, we used the averaged AQI score across the 3 closest monitoring stations.
PRIMARY ANALYSIS
We studied the association between AQI and the WHO Clinical Progression Scale score on day 28 following ICU admission. From the primary analysis, a 10-unit increase in the average AQI value over the past 12 months prior to COVID-19 diagnosis was associated with reduced odds of having a higher WHO Clinical Progression Scale score on the 28th day following ICU admission (OR 0.84; 95% CI 0.72–0.97, p = 0.02). Age (OR 1.40; 95% CI 1.32–1.49, p < .001), BMI (OR 1.03; 95% CI 1.02–1.04, p < .001), and Elixhauser indices for complicated hypertension (OR 0.64; 95% CI 0.44–0.94, p = 0.02), chronic pulmonary disease (OR 1.43; 95% CI 1.13–1.81, p = 0.003), renal failure (OR 2.06; 95% CI 1.52–2.80, p < .001), and liver disease (OR 1.68; 95% CI 1.29–2.19, p < .001) were independently associated with the WHO Clinical Progression Scale score. The model displayed moderate discriminative ability (concordance statistic = 0.67). Full results of the primary analysis can be found in Table 2.
Table 2. The Effect of Air Quality on Outcomes of COVID-19 Patient after ICU Stay
| Variables |
Adjusted Odds Ratio (95% CI)* |
P-Value |
| Average AQI in the Year Prior to COVID-19 Diagnosis (per 10-unit increase) |
0.84 (0.72, 0.97) |
0.019 |
| Age (per 10-year increase) |
1.40 (1.32, 1.49) |
<.001 |
| Body Mass Index |
1.03 (1.02, 1.04) |
<.001 |
| Congestive Heart Failure |
0.83 (0.65, 1.07) |
0.143 |
| Hypertension, Uncomplicated |
0.86 (0.71, 1.06) |
0.153 |
| Hypertension, Complicated |
0.64 (0.44, 0.94) |
0.021 |
| Chronic Pulmonary Disease |
1.43 (1.13, 1.81) |
0.003 |
| Diabetes, Uncomplicated |
0.89 (0.68, 1.17) |
0.402 |
| Diabetes, Complicated |
1.00 (0.76, 1.30) |
0.978 |
| Renal Failure |
2.06 (1.52, 2.80) |
<.001 |
| Liver Disease |
1.68 (1.29, 2.19) |
<.001 |
| Metastatic Cancer |
0.88 (0.55, 1.41) |
0.586 |
SECONDARY ANALYSES
We also studied the association between AQI and key secondary hospital outcomes. We found a 10-unit increase in average AQI was associated with an estimated increase of 0.83 (95% CI 0.17–1.50, p = 0.01) ventilation-free days, 0.88 (95% CI 0.12–1.63, p = 0.02) ICU-free days, and 0.89 (95% CI 0.17–1.61, p = 0.02) hospital-free days over 28-day period. Supplementary Tables S5, S6, and S7 contain full results of each respective analysis. We failed to detect an association between AQI and duration of hospitalization (p = 0.98) (Supplementary Table S8). In addition, a 10-unit increase in average AQI was associated with an estimated 19% reduced odds of in hospital mortality (OR 0.81; 95% CI 0.68–0.96, p = 0.02) (Supplementary Table S9). Lastly, a 10-unit increase in average AQI was associated with an estimated 24% decrease in the odds of being discharged to either a skilled nursing facility (OR 0.76; 95% CI 0.58–0.99, p = 0.04) or rehabilitation center (OR 0.76; 95% CI 0.60–0.96, p = 0.02), relative to being discharged home. Supplementary Table S10 contains the results of the discharge destination analyses.
SENSITIVITY ANALYSIS
As a sensitivity analysis, we examined the association between the WHO Clinical Progression Scale score and both the average AQI in the 2 months prior to COVID-19 diagnosis (OR 0.93; 95% CI 0.83–1.05, p = 0.26) (Table 3) and the average AQI in the 2 weeks prior to COVID-19 diagnosis (OR 0.95; 95% CI 0.87–1.05, p = 0.31) (Table 4). In contrast to the primary analysis, we failed to detect a significant association in either of these analyses.
Table 3. The Effect of Air Quality on Outcomes of COVID-19 Patient after ICU Stay
| Variables |
Adjusted Odds Ratio (95% CI)* |
P-Value |
| Average AQI in the 2 Months Prior to COVID-19 Diagnosis (per 10-unit increase) |
0.93 (0.83, 1.05) |
0.264 |
| Age (per 10-year increase) |
1.40 (1.32, 1.48) |
<.001 |
| Body Mass Index |
1.03 (1.02, 1.04) |
<.001 |
| Congestive Heart Failure |
0.83 (0.64, 1.06) |
0.131 |
| Hypertension, Uncomplicated |
0.88 (0.72, 1.07) |
0.193 |
| Hypertension, Complicated |
0.62 (0.43, 0.91) |
0.013 |
| Chronic Pulmonary Disease |
1.44 (1.14, 1.82) |
0.002 |
| Diabetes, Uncomplicated |
0.88 (0.67, 1.16) |
0.376 |
| Diabetes, Complicated |
1.00 (0.77, 1.31) |
0.995 |
| Renal Failure |
2.06 (1.52, 2.80) |
<.001 |
| Liver Disease |
1.67 (1.28, 2.18) |
<.001 |
| Metastatic Cancer |
0.86 (0.54, 1.38) |
0.527 |
Table 4. The Effect of Air Quality on Outcomes of COVID-19 Patient after ICU Stay
| Variables |
Adjusted Odds Ratio (95% CI)* |
P-Value |
| Average AQI in the 2 Weeks Prior to COVID-19 Diagnosis (per 10-unit increase) |
0.95 (0.87, 1.05) |
0.308 |
| Age (per 10-year increase) |
1.40 (1.32, 1.48) |
<.001 |
| Body Mass Index |
1.03 (1.02, 1.04) |
<.001 |
| Congestive Heart Failure |
0.83 (0.65, 1.06) |
0.140 |
| Hypertension, Uncomplicated |
0.88 (0.72, 1.07) |
0.196 |
| Hypertension, Complicated |
0.63 (0.43, 0.91) |
0.014 |
| Chronic Pulmonary Disease |
1.44 (1.14, 1.82) |
0.002 |
| Diabetes, Uncomplicated |
0.88 (0.67, 1.16) |
0.365 |
| Diabetes, Complicated |
1.00 (0.77, 1.31) |
0.990 |
| Renal Failure |
2.06 (1.51, 2.80) |
<.001 |
| Liver Disease |
1.66 (1.27, 2.16) |
<.001 |
| Metastatic Cancer |
0.87 (0.54, 1.39) |
0.558 |
DISCUSSION
We investigated whether air quality, measured using AQI, was associated with higher disease severity for critically ill patients with COVID-19. We found higher AQI was associated with decreased COVID-19 illness severity, measured using the WHO Clinical Progression Scale, at one month following ICU admission within our cohort. This finding may suggest that, within a respiratory pandemic, increased pollution of the environment could lead to lower illness severity. However, this finding is notable only when considering the average AQI for the year prior to admission. When averaging AQI 2 weeks or 2 months prior to admission, there was no significant correlation with illness severity. Together these findings contribute to literature by demonstrating AQI is likely a poor measure of clinically significant respiratory pollution and should be used with caution when examining the impact of air pollution on patient outcomes.
Air pollution is a significant environmental hazard that has implications on patient health.8 The United States Environmental Protection Agency defines a good AQI as values less than 50, with AQI calculations based on five major criteria pollutants regulated by the Clean Air Act (ozone, particle pollution, carbon monoxide, sulfur dioxide, and nitrogen dioxide; www.airnow.gov/). Our median AQI score was 37 (33–41). For comparison, the city of Shanghai had an average AQI of 56.5 (16–163) between January and March of 2020.13 There has been data within higher AQI cohorts demonstrating an association of higher AQI on increased daily COVID-19 cases and rates of death.14 While is possible that the severity of pollution within our cohort was not sufficient to pose heightened risk in at least 75% of our study subjects, our findings may be more generalizable to United States populations where average AQI in 2021 was 39.9 (www.airnow.gov/). In addition, our data are similar other studies in which older patients with higher BMI and with increased Elixhauser comorbidities were more likely to suffer prolonged severe COVID-19 disease course,15 demonstrating fidelity within our dataset.
While we are unable to draw conclusions on the association of AQI on COVID-19 disease severity, our study raises important questions toward understanding the interplay of environmental data on patient outcomes. PM2.5 and ozone, components of the AQI, have shown the greatest association to COVID-19 illness severity.16 These particular components have physiologic-driven damage pathways with PM2.5 inducing autophagy within lung tissues17 and ozone inflicting damage via reactive oxygen species mechanism.18 Our findings may be partially explained by the non-specificity of the AQI with certain components offsetting others. Geographic-driven pollution studies on air pollution and effects on long-term medical conditions have historically used exposure using to roads, fixed-site monitoring stations, land use regression and dispersion modelling, or individual residential levels.9 Regions and populations within our cohort exposed to worse AQI may have been impacted by these additional unmeasured geographic modifiers that could explain our findings that we could not capture. While presenting data with AQI, our findings suggest important considerations that may interact AQI and outcomes could confound findings.
Our study has several strengths. We used laboratory-confirmed COVID-19 disease subjects to ensure our population could be accurately assessed. Inclusion of patients with positive test results across the pandemic duration also improves generalizability toward later waves of the COVID-19 pandemic where our caseload was particularly increased. We used the validated WHO Clinical Progression Scale to demonstrate clinically relevant disease severity across our outcomes. Our large robust database strengthened by our underlying informatics infrastructure allowed for better accuracy of clinical outcomes. Our study also has several limitations. While the WHO Clinical Progression Scale score was designed to measure the severity of illness for COVID-19 patients, the principles we adopted for mapping the EHR data to WHO score may be imperfect. Second, it was challenging to determine how to deal with missing data values in air quality data management. We had a measuring station in 52% of our population and the rest of the data was estimated based on the closest three stations and their average. However, this approach could have led to inaccurate assessments of the exposure to poor quality air and high concentrations of hazardous air-pollutants. Similarly, air quality stations measurements rest on the differences between neighborhoods within the same region. Studies have shown that red-lined areas are up to 7 °C hotter than non-redlined areas despite being within the same city.19 This increase in heat can be attributed to the lack of tree canopy and more densely packed neighborhood structures resulting in decreased air circulation, increased air pollution, and increased heat.19 While patient address was taken into account, it is possible that the closest air quality station might average the difference of pollution in redlined and non-redlined neighborhoods resulting in an inaccurate AQI. We contend that the use of our approach was thoroughly considered in terms of imputation accuracy and computational feasibility, despite this, imputations could bias our findings on the associations between air quality and health outcomes. Due to the retrospective, observational nature of this study, we were unable to follow up on patients who were discharged home prior to the 28th day after ICU admission, which is problematic and could leads to bias. In addition, other social determinants of health – including poverty, physical environment (e.g., smoke exposure, homelessness), and race or ethnicity – were not included in this study, while they are believed to have a considerable effect on COVID-19 outcomes.20 The protective effect of AQI observed could potentially be related to an unmeasured, external factor.
CONCLUSIONS
In summary AQI appears to be a poor marker for qualifying the clinically significant impact of respiratory pollution on outcomes among critically ill COVID-19 patients. Our findings suggest important considerations should be made when investigating the impact of air pollution measured using the AQI on patient outcomes. Future studies are needed to explore more accurate markers for air quality and disease association and to better understand the interaction effects of different social determinants of health on patient outcomes.
REFERENCES
- Office of Disease Prevention and Health Promotion. Healthy People 2030 Framwork. HealthyPeople.gov. Published online 2018.
- Vazquez Guillamet MC, Dodda S, Liu L, et al. Race does not impact sepsis outcomes when considering socioeconomic factors in multilevel modeling. Crit Care Med 2022;50(3).
- Falvey JR, Murphy TE, Leo-Summers L, et al. Neighborhood socioeconomic disadvantage and disability after critical illness. Crit Care Med 2021; Publish Ah:1–9.
- Hsu HE, Ashe EM, Silverstein M, et al. Race/ethnicity, underlying medical conditions, homelessness, and hospitalization status of adult patients with covid-19 at an urban safety-net medical center — boston, Massachusetts, 2020. MMWR Morb Mortal Wkly Rep 2020;69(27):864–9.
- Penman-Aguilar A, Talih M, Huang D, et al. Measurement of health disparities, health inequities, and social determinants of health to support the advancement of health equity. J Public Heal Manag Pract 2016;22:S33–S42.
- Khreis H, Kelly C, Tate J, et al. Exposure to traffic-related air pollution and risk of development of childhood asthma: A systematic review and meta-analysis. Environ Int 2017;100.
- Proctor RN. The history of the discovery of the cigaretteelung cancer link: Evidentiary traditions, corporate denial, global toll. Tob Control 2012;21(2).
- Becchetti L, Beccari G, Conzo G, et al. Air quality and COVID-19 adverse outcomes: Divergent views and experimental findings. Environ Res 2021;193.
- WHO Working Group onthe Clinical Characterisation and Management of COVID-10 infection. A minimal common outcome measure set for COVID-19 clinical research. Lancet Infect Dis 2020;20(8).
- von Elm E, Altman DG, Egger M, et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Lancet Oct 20 2007;370(9596):1453–7.
- Ramaswamy J, Gong JJ, Saleh SN, et al. Developing a COVID-19 WHO Clinical Progression Scale inpatient database from electronic health record data. J Am Med Inform Assoc 2022;29(7):1279–85.
- Bellomo R, Ronco C, Kellum JA, et al. Acute Dialysis Quality Initiativew: Acute renal failure – definition, outcome measures, animal models, fluid therapy and information technology needs: the Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit Care 2004;8:R204–12.
- Tong L, Ji L, Li D, et al. The occurrence of COVID-19 is associated with air quality and relative humidity. J Med Virol 2022 Mar;94(3):965–70.
- Vali M, Hassanzadeh J, Mirahmadizadeh A, et al. Effect of meteorological factors and Air Quality Index on the COVID-19 epidemiological characteristics: an ecological study among 210 countries. Environ Sci Pollut Res Int 2021 Oct;28(38):53116–26.
- Richardson S, Hirsch JS, Narasimhan M, et al.; the Northwell COVID-19 Research Consortium. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City Area. JAMA 2020 May 26;323(20):2052–9.
- Chen Z, Sidell MA, Huang BZ, et al. Ambient air pollutant exposures and covid-19 severity and mortality in a cohort of patients with COVID-19 in Southern California. Am J Respir Crit Care Med 2022 Aug 15;206(4):440–8.
- Falcon-Rodriguez CI, Osornio-Vargas AR, Sada-Ovalle I, et al. Aeroparticles, composition, and lung diseases. Front Immuno 2016 Jan 20;7:3.
- Valavanidis A, Vlachogianni T, Fiotakis K, et al. Pulmonary oxidative stress, inflammation and cancer: respirable particulate matter, fibrous dusts and ozone as major causes of lung carcinogenesis through reactive oxygen species mechanisms. Int J Environ Res Public Health 2013 Aug 27;10(9):3886–907.
- Hoffman JS, Shandas V, Pendleton N. The Effects of historical housing policies on resident exposure to intra-urban heat: a study of 108 US Urban Areas. Climate 2020;8(1):12.
- Abrams EM, Szefler SJ. COVID-19 and the impact of social determinants of health. Lancet Respir Med 2020 Jul;8(7):659–61.
Article citation: Boncyk CS, Shirali S, Flores-Conner N, Li G, Clifton JC, Butler P, McCarthy KY, Freundlich RE. Examining the association of social determinants of health on critically ill COVID-19 patient outcomes: A retrospective observational study. The Southwest Journal of Medicine 2025;13(56):8–16
From: Vanderbilt University Medical Center, Nashville, TN (CSB, NF-C, GL, JCC, PB, KYM, REF) Eastern Virginia Medical School, Norfolk, VA (SS)
Submitted: 5/29/2025
Accepted: 6/17/2025
Conflicts of interest: Gen Li owns stock in Johnson & Johnson and Roche Holding AG. Dr. Freundlich owns stock in 3M and has received consulting fees from Phillips Healthcare and Oak Hill Clinical Informatics. Dr. Boncyk is a consultant for Sedana Medical. The other authors declare no competing interests.
This work is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.