INTRODUCTION
Sepsis and septic shock are caused by a dysregulated response to infection, leading to endothelial dysfunction, persistent hypotension, and life-threatening organ dysfunction.1 Septic shock remains a leading cause of death in intensive care units, with mortality rates estimated between 35% and 50%.2,3 The Surviving Sepsis Campaign Guidelines recommend prompt administration of broad-spectrum antibiotics and early intravenous fluid resuscitation as core elements of initial management.4 However, only about 50% of patients respond to fluid challenges, and hemodynamic benefits are often transient.5 As a result, vasopressors are frequently required to maintain adequate mean arterial pressures and organ perfusion. Norepinephrine is the first-line treatment, but escalating doses can result in adverse effects, including tachyarrhythmias, myocardial dysfunction, hyperglycemia, and even immunosuppression.6,7
Given these limitations, interest has grown in “multimodal” strategies that incorporate agents targeting different pathways.8 One such agent is methylene blue (MB, C16H18ClN3S, mass 319.85 g·mol−, see molecular structure below, Figure 1), which targets the nitric oxide (NO) signaling pathway, a key mediator of vasodilation in septic shock. Inflammatory cytokines and endotoxins induce overexpression of inducible nitric oxide synthase (iNOS), leading to excess NO production, which contributes to the pathologic vasodilation and reduced vascular tone characteristic of septic shock.9,10 Several non-selective NOS inhibitors, such as L-NMMA, have been studied in septic shock. While these drugs can increase mean arterial pressure and systemic vascular resistance, they also interfere with NO’s critical role in immune response. This may explain why clinical trials using NOS inhibitors have not reported improved outcomes, and in some cases have shown harm.11–13
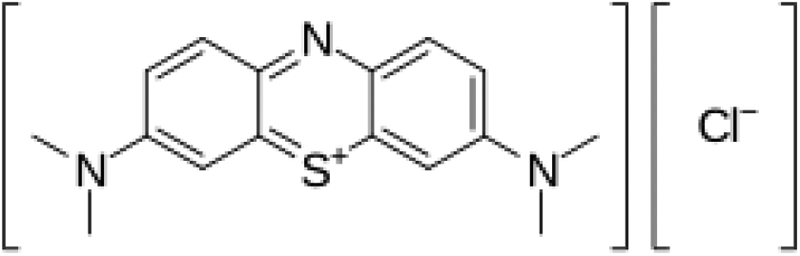
Figure 1. Molecular structure of methylene blue.
Methylene blue acts downstream of NO by inhibiting soluble guanylate cyclase (sGC), an enzyme responsible for converting NO signals into cyclic GMP (cGMP), which promotes vascular smooth muscle relaxation. By blocking sGC, MB reduces cGMP production and counteracts the vasodilatory effects of NO without completely suppressing its upstream immunologic functions.14,15 This targeted mechanism has led to MB’s use in other vasoplegic states, including post-cardiac surgery vasoplegia, anaphylaxis, and reperfusion syndrome following liver transplantation.16–18 This review summarizes the current literature on the physiologic and clinical effects of MB in septic shock.
PHYSIOLOGIC OUTCOMES
Ten studies with 296 patients were included in this review (Table 1). Methylene blue administration consistently improved hemodynamic parameters. An increase in MAP was the most reproducible finding across studies, supporting MB’s ability to restore vascular tone in septic shock.19–26 This effect was frequently accompanied by an increased systemic vascular resistance index (SVRI).19,21–24 Importantly, these changes occurred without significant alterations in cardiac output or cardiac index, suggesting that MB enhances perfusion pressure primarily through effects on peripheral resistance, without adversely impacting cardiac function.20,22–24 In some cases, left ventricular stroke work index (LVSWI) improved following MB administration, possibly as a secondary effect of improved vascular resistance and myocardial efficiency.20,23,24 A few studies also reported a transient decrease in serum lactate, which may reflect enhanced tissue perfusion, although this effect was not universal.20,24
Several studies also noted an increase in pulmonary vascular resistance and mean pulmonary artery pressure following MB administration.19,23,24 However, gas exchange was largely unaffected, and no studies reported clinically significant deterioration in respiratory status. A few early studies also observed a modest decline in PaO2/FiO2 ratio, but this was not reproduced in more recent RCTs.19,23,26 Collectively, these findings support MB’s ability to restore vascular tone in septic shock without compromising oxygen delivery or cardiac performance.
CLINICAL OUTCOMES
While the physiologic effects of MB are well documented, clinical outcomes remain inconsistent. Three studies observed a reduction in vasopressor requirements with MB administration.25,27,28 Ibrarra-Estrada et al. also reported shorter intensive care unit (ICU) and hospital lengths of stay.28 However, MB administration was not associated with a consistent survival benefit, and most studies found no statistically significant difference in mortality between MB and control groups.26–28 In the few studies that did report higher survival with MB, sample sizes were small and not adequately powered to detect true differences in outcomes.
Data on other clinical endpoints, such as mechanical ventilation duration, organ failure, and ICU-acquired complications, were also limited and inconsistently reported. Overall, MB appears to be a physiologically effective adjunct in the treatment of septic shock, but the current body of evidence does not yet support a definitive improvement in survival or other major clinical outcomes, highlighting the need for larger, well-powered randomized controlled trials.
DISCUSSION
Methylene blue demonstrates consistent improvements in hemodynamic parameters such as MAP and SVR without compromising cardiac output or oxygen delivery. These effects align with MB’s known inhibition of the NO-sGC-cGMP pathway. However, whether these physiologic outcomes translate into consistent clinical benefits remains uncertain. While MB frequently reduced vasopressor requirements and, in some studies, shortened ICU or hospital stay, evidence for improvement in mortality or other long-term endpoints is lacking.
The optimal timing for MB administration also remains under debate. In most studies, MB was used as a salvage therapy after conventional therapy had failed. Only one study investigated early use, administering MB within 24 hours of septic shock diagnosis.28 Emerging literature suggests there may be a therapeutic window early in the course of shock during which iNOS and sGC expression are elevated, making the vasculature more responsive to MB’s effects.29–31 Administering MB during this period may reverse vasoplegia more effectively, reducing catecholamine exposure and improving organ perfusion before metabolic failure ensues. In contrast, late-stage administration, often in the setting of sustained tissue hypoperfusion and lactic acidosis, may offer little benefit.
Dosing strategies for MB also varied considerably across studies. A bolus dose of 1 mg/kg was enough to improve vascular tone in many patients,22 although other studies have used doses up to 4 mg/kg or continuous infusions over 6 hours. Higher cumulative doses (>7 mg/kg) raise concerns for toxicity, including splanchnic ischemia.15 Nonetheless, MB was generally well tolerated, with most adverse events related to blue-green discoloration of urine, skin, and secretions, which may interfere with the accuracy of pulse oximeter readings.25,28 Paradoxical methemoglobinemia was observed in some patients,26,28 but it remained lower than the clinically relevant threshold of 10 percent in all studies. Methylene blue is contraindicated in patients with glucose-6-phosphate dehydrogenase (G6PD) deficiency due to risk of hemolysis, and inhibition of monoamine oxidase A by MB can induce serotonin syndrome in patients taking serotonergic medications.14 There were initial concerns regarding MB’s potential to impair pulmonary oxygen exchange or cause pulmonary vasoconstriction, reported in two early studies by Gachot et al. and Weingartner et al.19,23 However, this has not been substantiated in recent trials.26,28
Several meta-analyses of MB studies in shock have also been published, but all are based on very limited data. In a meta-analysis of 6 randomized controlled trials with a total of 302 patients, Fernando et al. found that MB was associated with reduced short-term mortality (RR 0.66, 95% CI 0.47–0.94), shortened vasopressor duration, and reduced hospital length of stay.32 Ng et al. analyzed five studies (257 patients) and found improvements in mean arterial pressure and oxygenation (PaO2/FiO2 ratio) but did not observe a statistically significant mortality benefit.33 Ballarin et al. analyzed three RCTs (141 patients) and reported that MB led to a significant decrease in ICU stay, fewer days on mechanical ventilation, and a faster time to vasopressor discontinuation, with no increase in adverse effects such as methemoglobinemia.34 The authors of these meta-analyses highlighted limitations that led to low certainty evidence, including the small sample sizes, heterogeneity of studies, and discoloration of skin/urine with MB administration that makes blinding difficult.
In summary, methylene blue shows promise as an adjunct therapy in septic shock, with the greatest potential in early intervention. Future research should focus on standardizing dosing regimens, clarifying optimal timing of administration, and identifying clinical or biochemical predictors of response. Larger randomized trials are needed to determine MB’s definitive place in the management of septic shock.
REFERENCES
- Singer M, Deutschman CS, Seymour CW, et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016;315(8):801–10. doi:10.1001/jama.2016.0287
- Bauer M, Gerlach H, Vogelmann T, et al. Mortality in sepsis and septic shock in Europe, North America and Australia between 2009 and 2019 – results from a systematic review and meta-analysis. Crit Care. 2020;24:239.
- Vincent JL, Jones G, David S, et al. Frequency and mortality of septic shock in Europe and North America: a systematic review and meta-analysis. Crit Care. 2019;23:196.
- Evans L, Rhodes A, Alhazzani W, et al. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock 2021. Intensive Care Med. 2021;47:1181–247.
- Bentzer P, Griesdale DE, Boyd J, et al. Will this hemodynamically unstable patient respond to a bolus of intravenous fluids? JAMA. 2016;316:1298–309.
- Russell JA. Vasopressor therapy in critically ill patients with shock. Intensive Care Med. 2019;45(11):1503–17. doi:10.1007/s00134-019-05801-z
- Stolk RF, Kox M, Pickkers P. Noradrenaline drives immunosuppression in sepsis: clinical consequences. Intensive Care Med. 2020;46:1246–8.
- Leone M, Einav S, Antonucci E, et al. Multimodal strategy to counteract vasodilation in septic shock. Anaesth Crit Care Pain Med. 2023;42(3):101193. doi: 10.1016/j.accpm.2023.101193
- Symeonides S, Balk RA. Nitric oxide in the pathogenesis of sepsis. Infect Dis Clin North Am. 1999;13(2):449-x. doi:10.1016/s0891-5520(05)70085-4
- Kirkebøen KA, Strand OA. The role of nitric oxide in sepsis–an overview. Acta Anaesthesiol Scand. 1999;43(3):275–88. doi:10.1034/j.1399-6576.1999. 430307.x
- Vincent JL, Zhang H, Szabo C, et al. Effects of nitric oxide in septic shock. Am J Respir Crit Care Med. 2000;161(6):1781–5. doi:10.1164/ajrccm.161.6.9812004
- Loughran PA, Lei Z, Xu L, et al. Nitric oxide in sepsis and hemorrhagic shock: beneficial or detrimental? In: Ignarro LJ, Freeman BA, editors. Nitric Oxide. 3rd ed. Cambridge: Academic Press; 2017. p. 289–300.
- Lopez A, Lorente JA, Steingrub J, et al. Multiple-center, randomized, placebo-controlled, double-blind study of the nitric oxide synthase inhibitor 546C88: effect on survival in patients with septic shock. Crit Care Med. 2004;32:21–30.
- Arias-Ortiz J, Vincent JL. Administration of methylene blue in septic shock: pros and cons. Crit Care. 2024;28(1):46.
- Jang DH, Nelson LS, Hoffman RS. Methylene blue for distributive shock: a potential new use of an old antidote. J Med Toxicol. 2013;9(3):242–9. doi:10.1007/s13181-013-0298-7
- Omar S, Zedan A, Nugent K. Cardiac vasoplegia syndrome: pathophysiology, risk factors and treatment. Am J Med Sci. 2015;349(1):80–8. doi:10.1097/MAJ.0000000000000341
- Mehaffey JH, Johnston LE, Hawkins RB, et al. Methylene blue for vasoplegic syndrome after cardiac operation: early administration improves survival. Ann Thorac Surg. 2017;104(1):36–41. doi: 10.1016/j.athoracsur.2017.02.057
- Hosseinian L, Weiner M, Levin MA, et al. Methylene blue: magic bullet for vasoplegia? Anesth Analg. 2016;122(1):194–201. doi:10.1213/ANE.0000000000001045
- Gachot B, Bedos JP, Veber B, et al. Short-term effects of methylene blue on hemodynamics and gas exchange in patients with septic shock. Intensive Care Med. 1995;21(10):1027–31. doi:10.1007/BF01700685
- Preiser JC, Lejeune P, Roman A, et al. Methylene blue administration in septic shock: a clinical trial. Crit Care Med. 1995;23(2):259–64. doi:10.1097/00003246-199502000-00008
- Daemen-Gubbels CR, Groeneveld AB, Thijs LG. Hemodynamic and metabolic effects of methylene blue in septic shock. Chest. 1995;108(4):1070–76. doi:10.1378/chest.108.4.1070
- Andresen M, Dougnac A, Diaz O, et al. Methylene blue in septic shock: a randomized, controlled, pilot study. Crit Care Med. 1998;26(12):1965–70. doi:10.1097/00003246-199812000-00007
- Weingartner R, Oliveira MG, Nunes DL, et al. Methylene blue in the treatment of refractory hypotension in distributive shock: a case series. Crit Care Med. 1999;27(9):2186–90. doi:10.1097/00003246-199909000-00021
- Donati A, Conti G, Loggi S, et al. Effects of methylene blue infusion on hemodynamics and oxygen transport in patients with septic shock. Crit Care Med. 2002;30(2):227–30. doi:10.1097/00003246-200202000-00002
- Kirov MY, Evgenov OV, Sovershaev MA, et al. Infusion of methylene blue in human septic shock: a pilot, randomized, controlled study. Crit Care Med. 2001;29(10):1860–7. doi:10.1097/00003246-200110000-00008
- Memis D, Turan A, Karamanlioglu B, et al. Methylene blue in septic shock: a randomized controlled study. Crit Care Med. 2002;30(5):1140–3. doi:10.1097/00003246-200205000-00013
- Rajbanshi LK, Singh D, Shrestha GS, et al. Effect of methylene blue on vasopressor requirements in patients with septic shock: a prospective study. J Nepal Health Res Counc. 2023;21(1):46–52. doi:10.33314/jnhrc. v21i1.4681
- Ibarra-Estrada MA, García-Salas Y, Mireles-Cabodevila E, et al. Adjunctive methylene blue for septic shock in the intensive care unit: a randomized controlled trial. J Crit Care. 2024;77:154375. doi: 10.1016/j.jcrc.2024.154375
- Evora PR. Methylene blue does not have to be considered only as rescue therapy for distributive shock. J Med Toxicol. 2013;9(4):426. doi:10.1007/s13181-013-0333-8
- Fernandes D, da Silva-Santos JE, Duma D, et al. Nitric oxide-dependent reduction in soluble guanylate cyclase functionality accounts for early lipopolysaccharide-induced changes in vascular reactivity. Mol Pharmacol. 2006;69(3):983–90. doi:10.1124/mol.105.015479
- Evora PR, Ribeiro PJ, Vicente WV, et al. Methylene blue for vasoplegic syndrome treatment in heart surgery: fifteen years of questions, answers, doubts and certainties. Rev Bras Cir Cardiovasc. 2009;24(3):279–88. doi:10.1590/s0102-76382009000400005
- Fernando SM, Alhazzani W, Burry L, et al. Methylene blue in patients with vasodilatory shock: a systematic review and meta-analysis of randomized trials. Crit Care Med. 2024;52(5):e274–e282. doi:10.1097/CCM.0000000000006265
- Ng B, Sidhu N, Bersten AD. Effect of methylene blue on outcomes in patients with vasoplegic shock: a systematic review and meta-analysis. J Crit Care. 2024;77:154334. doi: 10.1016/j.jcrc.2024.154334
- Ballarin A, Di Pasquale E, Candiani A, et al. Methylene blue in septic shock: a systematic review and meta-analysis. Braz J Anesthesiol. 2024. doi: 10.1016/j.bjane.2024.02.004
Article citation: Sang J, Alapati A, Nugent K. Outcomes of methylene blue treatment in septic shock. The Southwest Journal of Medicine 2025;13(56):25–29
From: School of Medicine (JS); Department of Internal Medicine (AA, KN), Texas Tech University Health Sciences Center, Lubbock, Texas
Submitted: 5/18/2025
Accepted: 7/14/2025
Conflicts of interest: none
This work is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.