Abstract
Frequency of peripheral arterial disease in patients presented with acute STEMI
Abdul Hameed MD, Shakir Zada MD, Ashan Ali Gnad MD, Zafar Iqbal MD, Barkha Daswani MD, Paras Nazir MD
Corresponding author: Abdul Hameed
Contact Information: Aamir09Lm@gmail.com
DOI: 10.12746/swjm.v13i57.1543
ABSTRACT
Background: Peripheral arterial disease (PAD) and coronary artery disease (CAD) are both manifestations of systemic atherosclerosis. In acute ST-elevation myocardial infarction (STEMI), the presence of PAD may reflect a more extensive vascular burden and worse clinical outcomes. The ankle–brachial index (ABI) is a simple, non-invasive tool to assess PAD, yet it remains underutilized in acute coronary care, particularly in high-risk populations such as STEMI patients.
Objective: To determine the frequency of PAD using ABI in patients presenting with acute STEMI and to evaluate its association with clinical outcomes and coronary lesion complexity.
Methods: A prospective cohort study was conducted at the National Institute of Cardiovascular Diseases, Karachi, over six months. A total of 246 patients aged 18–80 years, diagnosed with STEMI and undergoing percutaneous coronary intervention (PCI), were enrolled through non-probability consecutive sampling. ABI was measured bilaterally using Doppler and sphygmomanometer; values ≤0.90 indicated PAD. Data on demographics, comorbidities, echocardiographic findings, and angiographic parameters—including Syntax scores—were recorded. Statistical analysis was performed using SPSS version 21.0, with a p-value <0.05 considered significant.
Results: PAD was detected in 8.9% (right ABI) and 13.4% (left ABI) of STEMI patients. Diabetes was more prevalent in the PAD group (right ABI p = 0.039). PAD patients had lower ejection fraction (43.6 ± 7.7%), higher heart failure incidence (44.4%), and reduced PCI success rates (86.4%) compared to those without PAD. CABG planning was significantly higher in PAD-positive patients (40.9%, p = 0.042), though Syntax scores did not differ significantly across ABI groups (p = 0.592).
Conclusion: PAD is prevalent in STEMI patients and is associated with adverse clinical parameters and more intensive treatment strategies. ABI measurement offers a valuable addition to risk assessment and may guide more comprehensive cardiovascular management.
Keywords: Ankle–Brachial Index, Coronary Artery Disease, Diabetes Mellitus, Myocardial Infarction, Peripheral Arterial Disease, Risk Assessment, Vascular Diseases
INTRODUCTION
Peripheral arterial disease (PAD) remains an underrecognized yet significant manifestation of systemic atherosclerosis, particularly in patients presenting with acute coronary syndromes such as ST-elevation myocardial infarction (STEMI). While the focus of clinical management in STEMI has traditionally centered on rapid myocardial reperfusion and coronary artery stabilization, there is growing recognition that atherosclerosis is rarely confined to the coronary vasculature alone.1,2 PAD, characterized by narrowing or occlusion of the peripheral arteries—most commonly in the lower limbs—serves as a surrogate marker for widespread arterial disease. The coexistence of PAD in STEMI patients could signal a higher atherosclerotic burden and a potentially more complex clinical course. However, this vascular overlap remains insufficiently explored, particularly in low-to-middle income countries where both coronary artery disease (CAD) and PAD carry a high and often underestimated burden.3,4
The ankle–brachial index (ABI), a simple, non-invasive bedside test, has emerged as a reliable diagnostic tool for identifying PAD. An ABI value less than or equal to 0.9 is generally accepted as indicative of peripheral arterial obstruction.5,6 Numerous studies have demonstrated the prognostic value of ABI in predicting adverse cardiovascular outcomes, independent of other traditional risk factors such as diabetes, hypertension, and smoking. Yet, despite its ease of use and diagnostic utility, ABI is still not routinely assessed in acute cardiac care settings, especially in patients with STEMI, where the urgency of coronary intervention often sidelines peripheral vascular evaluation.7,8
Emerging data suggest that PAD is not merely a coexistent pathology but may be intricately linked with coronary lesion complexity and systemic atherosclerotic severity. Patients with PAD are more likely to harbor diffuse, multivessel CAD and may require more aggressive treatment strategies. Importantly, the presence of PAD could inform prognosis, guide therapeutic decisions, and potentially predict procedural complications during coronary interventions.9,10 This makes early identification of PAD in STEMI patients not only clinically relevant but potentially life-saving.11,12
Despite international attention to the systemic nature of atherosclerosis, there remains a noticeable gap in literature specifically quantifying the frequency of PAD among patients presenting with STEMI, particularly in high-volume tertiary cardiac centers.13 Most available evidence is derived from Western populations or focused on broader ACS cohorts, with limited attention to the unique clinical and demographic features of STEMI populations in South Asia. This study, conducted at the largest cardiac center in the world, seeks to address that gap by systematically evaluating the prevalence of PAD using ABI in patients presenting with acute STEMI.14
The rationale for this investigation lies in the premise that detecting PAD early in the STEMI population could serve as a practical marker for systemic atherosclerosis and help predict adverse cardiovascular outcomes. Therefore, the objective of this study is to determine the frequency of peripheral arterial disease, as identified by ABI, among patients presenting with acute ST-elevation myocardial infarction.
METHODS
This prospective cohort study was conducted at the National Institute of Cardiovascular Diseases (NICVD), Karachi, a high-volume tertiary cardiac center catering to a diverse patient population. The study spanned a duration of six months, commencing after formal approval from the institutional review board. Ethical standards were strictly adhered to throughout the research process, and verbal informed consent was obtained from all participants prior to data collection. Confidentiality and anonymity of patient data were maintained in compliance with ethical and institutional guidelines.15
Patients aged between 18 and 80 years, of either gender, presenting with acute ST-elevation myocardial infarction (STEMI) and undergoing percutaneous coronary intervention (PCI) for the first time were included using a non-probability consecutive sampling technique. Exclusion criteria comprised patients unwilling to provide consent and those with a documented history of previously treated coronary artery lesions, as such cases could potentially confound the evaluation of PAD prevalence in the context of a new STEMI event.16
Data were collected prospectively using a structured proforma designed to capture relevant clinical, laboratory, and angiographic parameters. Demographic variables such as age, gender, and residence were recorded, along with cardiovascular risk factors including hypertension, diabetes mellitus, smoking status, and personal or family history of coronary artery disease (CAD). Clinical presentation was classified based on ECG findings consistent with STEMI, and all patients underwent standard diagnostic workup including laboratory investigations, echocardiography, and coronary angiography.17
Peripheral arterial disease was assessed using the ankle–brachial index (ABI), calculated by dividing the systolic blood pressure at the ankle by that at the arm. A handheld Doppler device and standard sphygmomanometer were used to obtain the measurements. An ABI value of ≤0.9 was defined as indicative of PAD, while values >0.9 were considered normal. All patients underwent selective coronary angiography as per institutional protocols, but for this specific analysis, coronary lesion complexity (e.g., Syntax Score) was not the primary focus.18
Data entry and analysis were performed using IBM SPSS Statistics version 21.0 (IBM Corp., Armonk, NY, US). Continuous variables were analyzed for distribution; those normally distributed were expressed as means ± standard deviation, while skewed data were summarized using medians and interquartile ranges. Categorical variables were described as frequencies and percentages. The primary analysis involved estimating the frequency of PAD, as defined by ABI ≤0.9, among STEMI patients. Comparative analysis between PAD-positive and PAD-negative groups was performed using chi-square tests for categorical variables and independent-sample t-tests for continuous variables, as appropriate. A p-value of <0.05 was considered statistically significant for all analyses.19
This methodology aimed to ensure robust, transparent, and reproducible data collection to evaluate the burden of peripheral arterial disease in the context of acute ST-elevation myocardial infarction. However, one potential limitation lies in the use of verbal rather than written consent, which, while approved by the ethics board, may raise questions regarding documentation in certain regulatory settings. Additionally, although the ABI is a reliable bedside tool, variations in operator technique and patient hemodynamics during acute coronary events may influence measurement accuracy. These factors were minimized through standardization of procedure and training of personnel involved in data collection.20
RESULTS
Among the total of 246 patients presenting with acute ST-elevation myocardial infarction (STEMI), the frequency of peripheral arterial disease (PAD), defined by an ankle–brachial index (ABI) ≤0.90, was found to be 8.9% on the right side and 13.4% on the left side (Table 1). The proportion of patients with borderline ABI (0.91–1.4) was 83.7% (right) and 83.3% (left), while 7.3% (right) and 3.3% (left) of patients had ABI values >1.4, indicating potential non-compressible vessels.
Table 1. PAD Frequency by ABI Type
| ABI Type |
PAD Positive (≤0.90) |
Borderline (0.91–1.4) |
Non-compressible (>1.4) |
Total |
PAD Frequency (%) |
| Right ABI |
22 |
206 |
18 |
246 |
8.9% |
| Left ABI |
33 |
205 |
8 |
246 |
13.4% |
Among patients with right-sided ABI <0.90, 72.7% were male, and 50% had diabetes mellitus. The mean ejection fraction (EF) in this group was 43.6 ± 7.7%, which progressively declined across ABI categories, reaching 36.1 ± 8.3% in patients with ABI >1.4 (Figure 1). Heart failure incidence was also higher in the elevated ABI category, increasing from 22.7% in the low ABI group to 44.4% in the >1.4 group (Figure 2). Successful percutaneous coronary intervention (PCI) was achieved in 86.4% of patients with ABI <0.90, while the success rate was higher in the normal ABI group (98.1%) and 94.4% in the >1.4 group (p = 0.011). Diabetes mellitus showed a significant association with PAD (p = 0.039), especially in the ABI >1.4 group (72.2%) (Table 2).
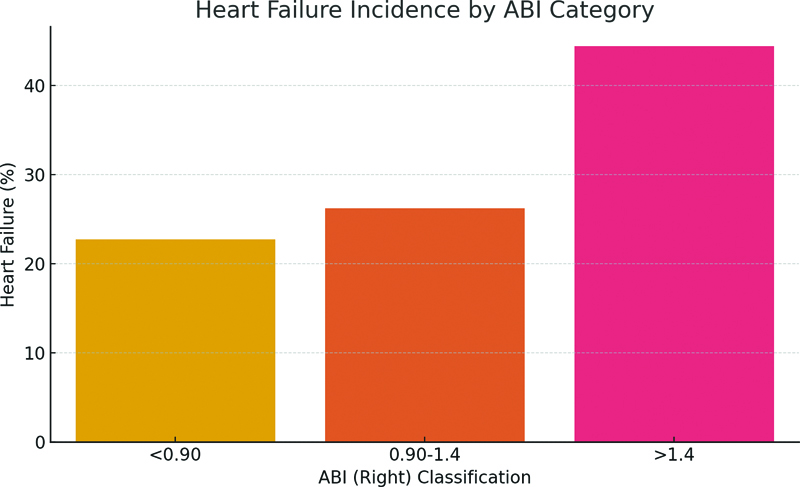
Figure 1. Heart failure incidence by ABI Category.
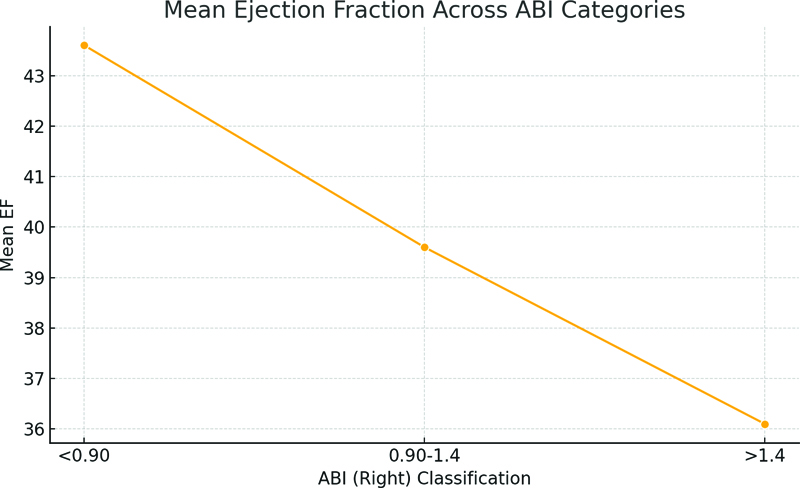
Figure 2. Mean Ejection fraction Across ABI Categories.
Table 2. Clinical Outcomes in PAD vs Non-PAD (Right ABI)
| Variable |
PAD Group (ABI ≤0.90) |
Non-PAD Group (ABI >0.90) |
| Heart Failure (%) |
22.7 |
26.7 |
| Successful PCI (%) |
86.4 |
97.6 |
| CABG Planned (%) |
40.9 |
25.0 |
| EF (Mean ± SD) |
43.6 ± 7.7 |
39.3 ± 8.7 |
| Mortality (%) |
4.5 |
2.4 |
Mean Syntax Scores were 25.8 ± 9.6, 23.4 ± 10.4, and 23.2 ± 9.6 in patients with ABI <0.90, 0.90–1.4, and >1.4 respectively, though no significant correlation was observed between ABI and Syntax classification (p = 0.592). Similarly, multivessel disease was common across all ABI groups (90.9% in ABI <0.90, 79.6% in ABI 0.90–1.4, and 88.9% in ABI >1.4), but did not differ significantly (p = 0.301). Left main coronary artery disease was present in 27.3% of patients with ABI <0.90 and in 11.1% of those with ABI >1.4 (p = 0.42) (Table 3).
Table 3. PAD Frequency across Syntax Score Categories
| Syntax Category |
Right ABI PAD Cases (≤0.90) |
Left ABI PAD Cases (≤0.90) |
Total in Group |
Right ABI PAD Frequency (%) |
Left ABI PAD Frequency (%) |
| Low (≤22) |
6 |
12 |
108 |
5.6% |
11.1% |
| Intermediate (23–32) |
13 |
15 |
98 |
13.3% |
15.3% |
| High (>32) |
3 |
6 |
40 |
7.5% |
15.0% |
The need for coronary artery bypass grafting (CABG) was higher in the ABI <0.90 group (40.9%) compared to 5.6% in the >1.4 ABI group (p = 0.042) (Table 2). Mortality was recorded in 7 patients (2.9%), distributed as 4.5%, 2.4%, and 5.6% across the ABI categories <0.90, 0.90–1.4, and >1.4 respectively, without significant difference (p = 0.661). Notably, left ventricular thrombus formation and mechanical circulatory support were more frequent in patients with elevated ABI (>1.4), though statistical significance varied.
A similar pattern was seen in left ABI classification: PAD (ABI ≤ 0.90) was detected in 33 patients (13.4%). The group with low ABI showed higher prevalence of diabetes (60.6%), heart failure (27.3%), and lower EF (38.5 ± 8.4%). Hemodialysis and mechanical support were significantly higher in this subgroup (p < 0.001). Patients with PAD also had higher rates of unsuccessful PCI (12.1%, p = 0.002) and longer hospital stays, as suggested by lower discharge-to-home percentages.
When stratified by Syntax score, 43.9% of the study population had low Syntax score (≤22), 39.8% intermediate (23–32), and 16.3% high (>32). PAD was more frequent in patients with intermediate and high Syntax scores (13.3% and 7.5% respectively) compared to those with low scores (5.6%), but the association did not reach statistical significance (p = 0.37 for right ABI). However, multivessel disease and left main involvement were significantly more common among patients with higher Syntax scores (p < 0.001) (Table 3).
Subgroup analysis by age showed that PAD was only observed in the 41–60 years group, with no cases in younger or older patients (Table 4). Similarly, analysis by ACS type revealed that PAD was most frequent in patients with AWMI and IWMI, while absent in HLWMI and IPWMI subtypes (Table 5). A detailed comparison of clinical variables across ABI categories is presented in Table 6.
Table 4. Subgroup Analysis: PAD by Age Group
| Age Group |
PAD Negative |
PAD Positive |
| <40 |
0 |
0 |
| 41–60 |
224 |
22 |
| >60 |
0 |
0 |
Table 5. Subgroup Analysis: PAD by ACS Type
| ACS Type |
PAD Negative |
PAD Positive |
| AWMI |
76 |
7 |
| HLWMI |
5 |
0 |
| IPWMI |
15 |
0 |
| IWMI |
55 |
7 |
| NSTEMI |
60 |
6 |
| USA |
13 |
2 |
Table 6. ABI Right Categories
| ABI Category |
Total Patients |
Male (%) |
Diabetes Mellitus (%) |
Heart Failure (%) |
Successful PCI (%) |
Mean EF |
Mean Syntax Score |
| <0.90 |
22 |
72.7 |
50 |
22.7 |
86.4 |
43.6 |
25.8 |
| 0.90–1.4 |
206 |
69.9 |
41.7 |
26.2 |
98.1 |
39.6 |
23.4 |
| >1.4 |
18 |
88.9 |
72.2 |
44.4 |
94.4 |
36.1 |
23.2 |
These findings highlight the relatively high frequency of PAD among acute STEMI patients and its association with adverse cardiovascular characteristics such as lower ejection fraction (Figure 2), higher CABG planning rates (Table 2), and increased need for mechanical support. Despite the absence of statistically significant associations with Syntax scores, the presence of PAD correlated with multiple clinical vulnerabilities.
DISCUSSION
The present study explored the frequency of peripheral arterial disease (PAD) among patients presenting with acute ST-elevation myocardial infarction (STEMI) and found that 8.9% of participants exhibited a right-sided ankle–brachial index (ABI) ≤0.90, while left-sided PAD was observed in 13.4% of cases. These findings highlight a notable prevalence of PAD in this high-risk cardiac cohort, supporting the concept that atherosclerosis is not limited to the coronary circulation but rather reflects a systemic vascular pathology. Although ABI remains underutilized in acute cardiac care, its diagnostic and prognostic value has been reinforced through this study by revealing clinically significant associations with cardiac dysfunction and procedural outcomes.
The observed PAD prevalence falls within the range reported in previous international literature, though slightly lower than in Western populations, where PAD in STEMI cohorts has reached 15–25% depending on the diagnostic threshold and population characteristics. Several factors may account for this variance, including younger age distribution, lifestyle differences, and underdiagnosed asymptomatic PAD in the South Asian population. However, the left-sided ABI values in this study indicated a higher frequency of PAD, which may reflect hemodynamic discrepancies or measurement variability, particularly in the setting of acute hemodynamic compromise. This reinforces the importance of bilateral ABI assessment, as unilateral measurements may underestimate systemic vascular involvement.21,22
In patients with PAD, significantly reduced ejection fraction and higher heart failure rates were documented, along with lower procedural success in percutaneous coronary intervention. These associations align with prior studies which have established that patients with ABI ≤0.90 often have multivessel and complex coronary artery disease, increased myocardial damage, and poorer hemodynamic stability during intervention. Although the present study did not demonstrate a statistically significant correlation between PAD and Syntax score classification, the numerical trend toward higher Syntax scores in PAD-positive patients suggests an underlying relationship that might have been more evident in a larger sample or with inclusion of additional clinical risk scoring systems. The elevated frequency of CABG planning among PAD patients further supports the notion of a more aggressive or diffuse atherosclerotic burden in this subgroup.23,24
One of the strengths of this study lies in its execution within the world’s largest dedicated cardiac center, allowing access to a substantial and clinically diverse population. The use of ABI as a non-invasive, easily reproducible tool added practical relevance to the findings, especially in a resource-constrained setting. Moreover, the direct focus on STEMI patients, as opposed to the broader ACS spectrum, allows for targeted insights into this high-risk and time-sensitive population.7,25
Despite these strengths, certain limitations merit consideration. The reliance on verbal consent, though ethically approved, may raise concerns in settings requiring formal documentation. Additionally, the cross-sectional nature of PAD assessment during the acute phase of STEMI may have been influenced by transient hemodynamic changes, potentially leading to measurement bias. ABI measurements are also operator-dependent and can be affected by arterial stiffness or non-compressible vessels, particularly in patients with diabetes or chronic kidney disease. The study did not include long-term follow-up, thus limiting the understanding of how PAD influenced post-discharge cardiovascular outcomes or mortality. Furthermore, the lack of multivariate regression analysis precluded identification of independent predictors of PAD in this cohort.16,26
The implications of these findings are clinically meaningful. Early recognition of PAD in STEMI patients can serve as a surrogate marker of systemic atherosclerosis and may assist in risk stratification, prognostication, and therapeutic decision-making. Incorporating ABI screening in emergency cardiac care could be particularly beneficial in resource-limited settings, where sophisticated diagnostic modalities may not be readily available. Future studies should aim to include larger, multicenter cohorts and explore the longitudinal impact of PAD on cardiovascular morbidity and mortality post-STEMI. Incorporating additional vascular markers and correlating with imaging or biochemical parameters may further refine the understanding of systemic vascular involvement in acute coronary syndromes.25,27
This study underscores the need to recognize PAD as an integral component of cardiovascular risk assessment in STEMI patients. The coexistence of lower extremity arterial disease with coronary pathology not only reflects widespread vascular compromise but also warrants a more comprehensive and anticipatory approach to cardiovascular care.
CONCLUSION
This study concluded that peripheral arterial disease is a frequently overlooked yet clinically relevant condition in patients presenting with acute ST-elevation myocardial infarction. The presence of PAD, as identified through ankle–brachial index assessment, reflected broader atherosclerotic involvement and was associated with adverse clinical features such as reduced cardiac function and lower procedural success. These findings emphasize the importance of incorporating PAD screening into routine evaluation of STEMI patients, not only to enhance diagnostic accuracy but also to support more informed and individualized treatment planning. Recognizing PAD in this context offers a simple, accessible, and meaningful way to improve cardiovascular risk stratification and overall patient care.
ACKNOWLEDGMENT
The authors sincerely thank the clinical and research teams at NICVD for their support and contribution.
REFERENCES
- Farkas K, Kolossváry E, Ferenci T, et al. Ankle brachial index is a strong predictor of mortality in hypertensive patients: results of a five-year follow-up study. Int Angiol. 2022;41(6):517–24.
- Liu YH, Tsai WC, Chi NY, et al. The association between percentage of mean arterial pressure and long-term mortality in acute myocardial infarction patients: an observational cohort study. Int J Med Sci. 2024;21(11):2119–26.
- Ban S, Sakakura K, Jinnouchi H, et al. Association of asymptomatic low ankle-brachial index with long-term clinical outcomes in patients after acute myocardial infarction. J Atheroscler Thromb. 2022;29(7):992–1000.
- Lilova Z, Hassan F, Riaz M, et al. Blood group and ischemic stroke, myocardial infarction, and peripheral vascular disease: a meta-analysis of over 145,000 cases and 2,000,000 controls. J Stroke Cerebrovasc Dis. 2023;32(8):107215.
- Brown T, Forster RB, Cleanthis M, et al. Cilostazol for intermittent claudication. Cochrane Database Syst Rev. 2021;6(6):Cd003748.
- Bechelli C, Macabrey D, Deglise S, et al. Clinical potential of hydrogen sulfide in peripheral arterial disease. Int J Mol Sci. 2023;24(12).
- Bonaca MP, Hamburg NM, Creager MA. Contemporary medical management of peripheral artery disease. Circ Res. 2021;128(12):1868–84.
- Wadström BN, Pedersen KM, Wulff AB, et al. Elevated remnant cholesterol and atherosclerotic cardiovascular disease in diabetes: a population-based prospective cohort study. Diabetologia. 2023;66(12):2238–49.
- Higashikuni Y, Liu W, Sata M. Give a leg up: screening for peripheral artery disease after acute myocardial infarction. J Atheroscler Thromb. 2022;29(7):989–91.
- Kridin K, Valido K, Cohen JM, et al. Hidradenitis suppurativa and the risk of myocardial infarction, cerebrovascular accident, and peripheral vascular disease: a population-based study. Arch Dermatol Res. 2023;315(3):429–35.
- Golledge J, Moxon JV, Rowbotham S, et al. High ankle brachial index predicts high risk of cardiovascular events amongst people with peripheral artery disease. PLoS One. 2020;15(11):e0242228.
- King A, Rajpura J, Liang Y, et al. Impact of cardiovascular disease on health care economic burden and resource utilization: a retrospective cohort study in adults in the United States with type 2 diabetes with or without stroke, myocardial infarction, and peripheral arterial disease. Curr Med Res Opin. 2022;38(11):1831–40.
- Abdel-Galeel A, El-Zokaim A, Hasan-Ali H, et al. Low ankle-brachial index is associated with microvascular coronary obstruction after primary PCI. Vasc Health Risk Manag. 2021;17:23–32.
- Bozkurt B, Savarese G, Adamsson Eryd S, et al. Mortality, outcomes, costs, and use of medicines following a first heart failure hospitalization: EVOLUTION HF. JACC Heart Fail. 2023;11(10):1320–32.
- Sigvant B, Hasvold P, Thuresson M, et al. Myocardial infarction and peripheral arterial disease: treatment patterns and long-term outcome in men and women results from a Swedish nationwide study. Eur J Prev Cardiol. 2021;28(13):1426–34.
- Luo J, Thomassen JQ, Nordestgaard BG, et al. Neutrophil counts and cardiovascular disease. Eur Heart J. 2023;44(47):4953–64.
- Annex BH, Cooke JP. New directions in therapeutic angiogenesis and arteriogenesis in peripheral arterial disease. Circ Res. 2021;128(12):1944–57.
- Mihatov N, Mosarla RC, Kirtane AJ, et al. Outcomes associated with peripheral artery disease in myocardial infarction with cardiogenic shock. J Am Coll Cardiol. 2022;79(13):1223–35.
- Sykora D, Firth C, Girardo M, et al. Patient age at diagnosis of peripheral artery disease and its impact on cardiovascular and limb outcomes. Am J Cardiol. 2022;177:144–50.
- Smolderen KG, Lee M, Arora T, Simonov M, Mena-Hurtado C. Peripheral Artery Disease and COVID-19 Outcomes: Insights from the Yale DOM-CovX Registry. Curr Probl Cardiol. 2022;47(12):101007.
- Shamaki GR, Markson F, Soji-Ayoade D, et al. Peripheral artery disease: a comprehensive updated review. Curr Probl Cardiol. 2022;47(11):101082.
- Rymer J, Anand SS, Sebastian Debus E, et al. Rivaroxaban plus aspirin versus aspirin alone after endovascular revascularization for symptomatic PAD: insights from VOYAGER PAD. Circulation. 2023;148(24):1919–28.
- Cicero AFG, Salvetti M. Peripheral artery disease: a highly prevalent untreated and uncontrolled independent cardiovascular disease. Atherosclerosis. 2022;354:55–6.
- Lingvay I, Brown-Frandsen K, Colhoun HM, et al. Semaglutide for cardiovascular event reduction in people with overweight or obesity: SELECT study baseline characteristics. Obesity (Silver Spring). 2023;31(1):111–22.
- Qin L, Chen B, Niu JY, et al. [The prevalence and risk factors of diabetic peripheral artery disease in Chinese communities]. Zhonghua Liu Xing Bing Xue Za Zhi. 2022;43(12):1932–8.
- Hess CN, Bonaca MP. Risk of remnants and remnant risk in peripheral artery disease. Eur Heart J. 2022;43(34):3270–2.
- Kaplovitch E, Eikelboom JW, Dyal L, et al. Rivaroxaban and aspirin in patients with symptomatic lower extremity peripheral artery disease: a subanalysis of the COMPASS randomized clinical trial. JAMA Cardiol. 2021;6(1):21–9.
Article citation: Hameed A, Zada S, Gnad AA, et al. Frequency of peripheral arterial disease in patients presented with acute STEMI. The Southwest Journal of Medicine 2025;13(57):22–29
From: National Institute of Cardiovascular Diseases, Karachi Pakistan (AH, SZ, ZI, BD, PN) Bahria University Health Sciences Campus, Karachi, Pakistan (AAG)
Submitted: 7/30/2025
Accepted: 9/19/2025
Conflicts of interest: none
This work is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.