Abstract
Silent culprit: Left atrial myxoma presenting as anterior STEMI
Ben Thiravetyan, MD, Dina Soliman, MD, Muhammad Qudrat Ullah, MD, Zhaunn Sly, MD
Corresponding author: Zhaunn Sly
Contact Information: Zhaunn.Sly@ttuhsc.edu
DOI: 10.12746/swjm.v14i58.1567
ABSTRACT
Primary cardiac tumors are extremely rare, with myxomas being the most common. While systemic embolization is a known complication of myxomas, coronary embolism is exceedingly rare. We report a case of a 70-year-old woman with anterior STEMI and no clear culprit lesion who was later found to have a large left atrial myxoma with mobile thrombus. This 70-year-old woman with hypertension, diabetes, and tobacco use presented with acute retrosternal chest pain. Initial ECG showed ST-segment elevation and Q waves in leads V2-V6. Coronary angiogram showed obstructive coronary arteries: 70% proximal LAD, 80% proximal-to-mid RCA, 43% distal LM. No clear culprit lesion to explain STEMI found by both angiography and intravascular imaging. A percutaneous coronary intervention was performed on the LAD and RCA with resolution of chest pain and improved, but persistent, ST elevation on ECG. On the next day, she developed recurrent chest pain; her ECG showed slightly increased ST elevation. Echocardiogram revealed EF 35% and a large myxoma with mobile thrombus attached. In this case, patient’s STEMI was suspected to be embolic in origin. Despite atherosclerotic risk factors and diffuse CAD on angiogram, no clear culprit lesion explained the persistent transmural ischemia seen on ECG, raising the possibility of heavy microvascular embolization.
Keywords: Myxoma; acute coronary syndrome; myocardial infarction
INTRODUCTION
Primary cardiac tumors are extremely rare, with a reported prevalence of approximately 0.02% based on multiple large autopsy series.1 Myxomas are the most common type of primary cardiac tumor, accounting for nearly half of all benign cardiac tumors.2 They are more frequently diagnosed in women between the third and sixth decades of life.2,3 Most myxomas originate from the interatrial septum and are predominantly located in the left atrium (approximately 75%), followed by the right atrium (approximately 15–20%), and rarely in the ventricles.2 Some myxomas are predominantly found in the left atrium, systemic embolization is a recognized complication.3 However, coronary embolization is extremely rare and poses a diagnostic challenge, as it can mimic or coexist with acute coronary syndrome due to atherosclerotic disease. We report a rare case of a 70-year-old woman presenting with anterior STEMI with unclear culprit lesion on coronary angiogram who was subsequently found to have a large left atrial myxoma with mobile thrombus.
CASE
A 70-year-old woman with a history of hypertension, diabetes, and tobacco use presented to the hospital with acute-onset retrosternal chest pain for three hours. The pain began while she was gardening and was described as constant, pressure-like, and associated with shortness of breath. After two hours without relief, she called emergency medical service (EMS), which administered 324 mg of aspirin and three doses of sublingual nitroglycerin, with minimal symptomatic improvement.
Upon arrival at the emergency department (ED), she was in moderate distress with persistent chest pain. She was hemodynamically stable (blood pressure (BP) 144/82 mmHg, heart rate (HR) 79 bpm) and was saturating well on room air. Physical exam was unremarkable. Her past medical history was significant for hypertension and diabetes, with medication nonadherence. She had no known prior cardiac history. Social history was remarkable for active tobacco use with a 10 pack-year smoking history. She denied alcohol or recreational drug use. Electrocardiogram (ECG) demonstrated sinus rhythm with ST-segments elevation and Q waves in leads V2–V6 (Figure 1A), consistent with an anterior ST-elevation myocardial infarction (STEMI). A rapid point of care echocardiogram revealed a mildly reduced left ventricular ejection fraction (LVEF) of 45–50% with anteroapical hypokinesis. Patient was started on high-intensity statin and heparin loading and infusion and was emergently taken to the cardiac cath lab.
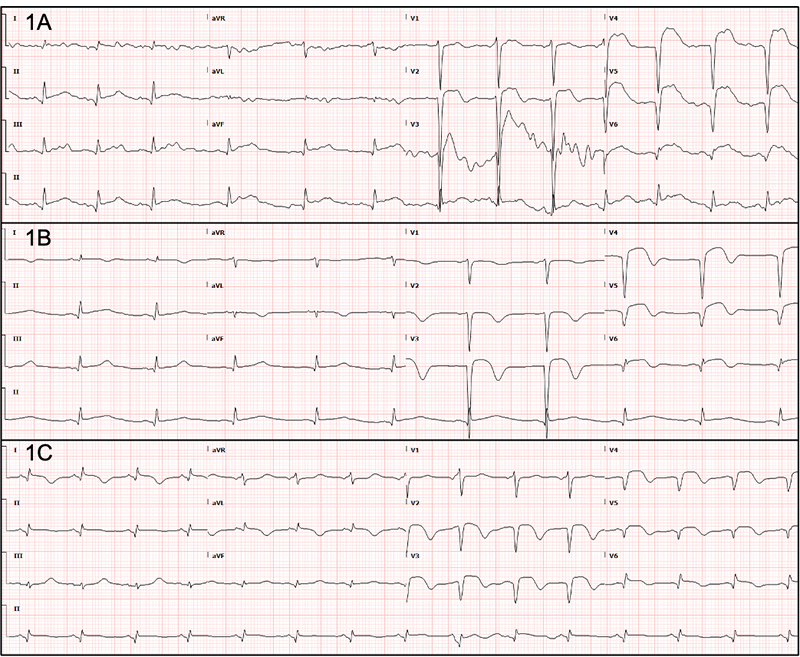
Figure 1. (A) ECG at presentation – Sinus rhythm with ST-segments elevation and Q waves in leads V2–V6. (B) ECG after PCI – Improvement in ST-segment elevation in leads V2–V6 with new T wave inversions in leads V2–V6, I, and aVL. (C) ECG on the next day – Increased ST-segment elevation in leads V2, I, and aVL with decreased voltage in leads II III, and aVF.
Emergent coronary angiography was performed via radial access. It revealed obstructive coronary artery disease (CAD) with 70% stenosis in the proximal left anterior descending artery (LAD), along with diffuse distal disease, and 80% stenosis in the proximal-to-mid segment of the right coronary artery (RCA) (Figure 2). The distal left main (LM) coronary artery showed 40–50% stenosis; however, intravascular ultrasound (IVUS) demonstrated a minimum luminal area of 7.8 mm2 with 43.1% stenosis (Figure 3). No unstable lesions were identified by intravascular imaging. Percutaneous intervention (PCI) was performed on the LAD using a 3.5 × 18 mm Xience drug-eluting stent (DES), with persistent TIMI-1 flow. The RCA underwent balloon angioplasty with a 2.5 × 20 mm compliant balloon, followed by placement of three Xience DES: 3.5 × 28 mm, 4 × 18 mm, and 3.5 × 8 mm.
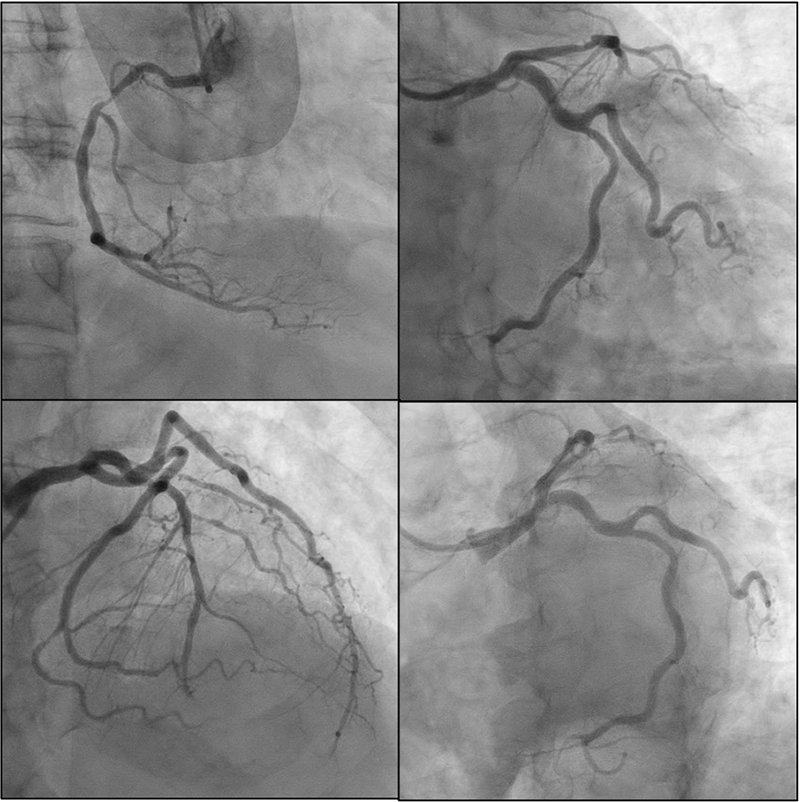
Figure 2. Coronary angiogram. Obstructive CAD with 70% stenosis in the proximal LAD, along with diffuse distal disease, 80% stenosis in the proximal-to-mid segment of the RCA, and 40–50% stenosis of distal LM.
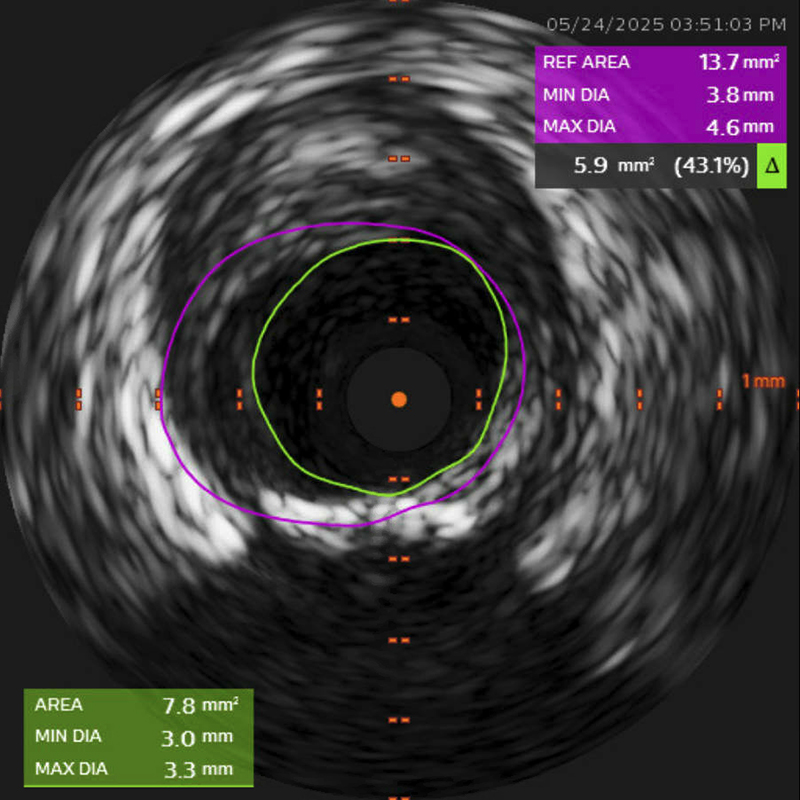
Figure 3. Intravascular ultrasound (IVUS). IVUS demonstrated a minimum luminal area of 7.8 mm2 with 43.1% stenosis of the distal left main coronary artery.
Following the procedure, the patient reported resolution of chest pain. She remained hemodynamically stable (BP 120/69 mmHg, HR 58 bpm) and continued to oxygenate well on room air. She was started on aspirin 81 mg daily, prasugrel 10 mg daily with loading, and 12-hour of tirofiban drip. Post-procedure ECG showed improvement in ST-segment elevation in leads V2-V6 with new T wave inversions in leads V2-V6, I, and aVL (Figure 1B). Labs came back remarkable for troponin T 1725 ng/L, brain natriuretic peptide (BNP) 1282 pg/mL, A1c 5.9%, LDL 93 mg/dL, and a urine drug screen positive for amphetamine.
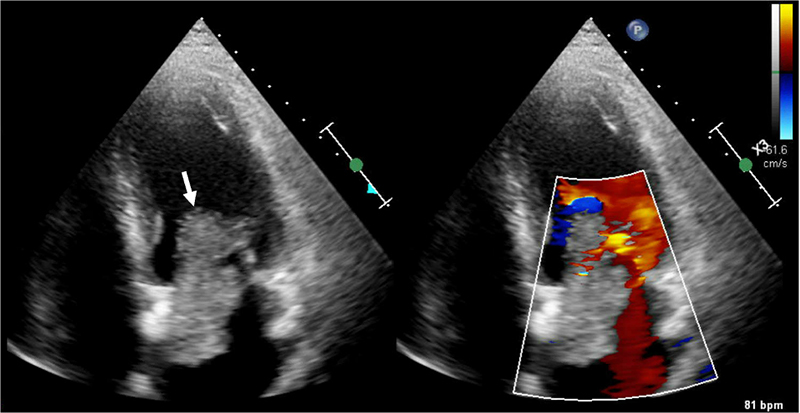
Figure 4. Transthoracic Echocardiogram. White arrow – 7.8 cm × 3.1 cm large echogenic structure in the left atrium attached to the interatrial septum protruding through mitral valve during diastole with mobile thrombus attached to the mass.
The next morning, patient experienced recurrent chest pain after returning from shower. She also reported bilateral leg heaviness and nausea, which partially improved with rest. However, her chest pain persisted, described as a pressure-like sensation worsened by deep inspiration. She remained hemodynamically stable (BP 130/87 mmHg, HR 83 bpm) but had increased oxygen requirement to 3 L/min via nasal cannula. Labs were remarkable for troponin T > 10000 ng/L. Repeat ECG showed increased ST-segment elevation in leads V2, I, and aVL with decreased voltage in leads II III, and aVF (Figure 1C). Transthoracic echocardiogram (TTE) demonstrated moderately reduced left ventricular systolic function (EF 35-39%), apical dyskinesis, anterior septal and mid anterior wall akinesis, mid lateral wall hypokinesis, reduced right ventricular systolic function, mild to moderate diastolic dysfunction, mild mitral regurgitation, and a 7.8 cm × 3.1 cm large echogenic structure in the left atrium attached to the interatrial septum protruding through mitral valve during diastole with mobile thrombus attached to the mass (Figure 4), suggestive of myxoma with superimposed thrombus.
Given high risk of thromboembolic complications, a heparin drip was started. A nitroglycerin drip was started for recurrent chest pain. Cardiothoracic surgery was emergently consulted given the high-risk myxoma features. Patient was considered she with an 80% ABG arterial blood gas high-risk for surgery due to her very recent anterior STEMI and reduced LVEF. Intra-aortic balloon pump (IABP) was offered as a salvage therapy for presumed no-reflow and/or microvascular thrombus of her coronary vasculature but was declined by the patient. She opted for medical management with dual antiplatelet therapy, heparin drip, and tirofiban drip, with the hope of achieving clinical stabilization to enable future surgical intervention.
CTS computed tomography with angiography patient remained hemodynamically stable and continued to require 3 L/min oxygen. However, six hours later, she suddenly developed agonal breathing and progressed into pulseless electrical activity (PEA) cardiac arrest. Advanced cardiac life support (ACLS) was initiated, but no return of spontaneous circulation (ROSC) was achieved. After 15 minutes of resuscitation efforts, family elected to discontinue CPR, and patient passed away.
DISCUSSION
The clinical presentation of myxomas varies depending on their size, location, mobility, and surface characteristics. They typically present as a triad of constitutional symptoms, embolic events, and intracardiac obstruction, although it is uncommon for all three manifestations to occur simultaneously.3,4 Villous-type myxomas are particularly friable and more prone to fragmentation, increasing the risk of embolization. Given their frequent occurrence in the left atrium, systemic embolization to the brain and peripheral extremities is a major concern.3 Embolization to the coronary arteries is exceedingly rare and there are only a few case reports available in the literature.5–7
The team was concerned that the presenting STEMI could have been embolic in origin from the thrombus on the left atrial myxoma which was later discovered the next day. Although patient had multiple atherosclerotic risk factors, including chronic tobacco use, poorly controlled hypertension, postmenopausal status, and questionable amphetamine use, and coronary angiogram revealed evidence of diffuse atherosclerosis in multiple vessels, there was no clearly identifiable culprit lesion that would explain the persistent transmural ischemia seen on the ECG. One possible explanation may be heavy burden microvascular obstruction from cardiac embolism in the setting of underlying advanced coronary artery disease.
In this case, patient presented with an acute anterior STEMI. However, the presence of Q waves in the anterior leads suggested a subacute infarction. Given her ongoing chest pain, patient was taken emergently for revascularization in an attempt to preserve the remaining viable myocardium. After PCI to the LAD and RCA, patient was free of pain. Nonetheless, ECG continued to show persistent ST segments elevation and Q waves which raised concerns for myocardial scarring or aneurysm. In addition, despite extensive infarct area and significant regional wall abnormalities, patient’s hemodynamic remained stable, which also suggested that some degree of the cardiac dysfunction may be chronic.
The cause of patient’s sudden PEA cardiac arrest remained unclear. Possible etiologies included acute obstruction of mitral valve by myxoma causing sudden hemodynamic collapse, large embolic stroke involving brainstem, large coronary emboli leading to acute infarction on top of a recent STEMI, or tumor rupture with acute hemopericardium and tamponade.
Another important learning point from this case is the diagnostic value of bedside echocardiogram. While STEMI is an emergent condition that warrant immediate revascularization as “time is muscle,” a bedside echocardiogram should not only focus on evaluating the LVEF and wall motion abnormalities. Physicians should also assess for non-atherosclerotic causes of acute myocardial infarction, such as aortic dissection or cardiac tumor.
It would be easier for me to retake this case highlights the importance of considering alternative causes of STEMI, particularly in the absence of a clear culprit lesion on coronary angiogram, and the importance of bedside echocardiogram in evaluating for structural abnormalities in patient presenting with STEMI. Left atrial myxoma, although rare, can lead to coronary embolism and presented as STEMI.
REFERENCES
- Reynen K. Frequency of primary tumors of the heart. Am J Cardiol 1996;77(1):107. (In eng). DOI: 10.1016/s0002-9149(97)89149-7.
- Reynen K. Cardiac myxomas. N Engl J Med 1995;333(24):1610–17. (In eng). DOI: 10.1056/nejm199512143332407.
- Pinede L, Duhaut P, Loire R. Clinical presentation of left atrial cardiac myxoma. A series of 112 consecutive cases. Medicine (Baltimore) 2001;80(3):159–72. (In eng). DOI: 10.1097/00005792-200105000-00002.
- Griborio-Guzman AG, Aseyev OI, Shah H, et al. Cardiac myxomas: clinical presentation, diagnosis and management. Heart 2022;108(11):827–33. (In eng). DOI: 10.1136/heartjnl-2021-319479.
- Peters MJ, Tuwairqi KW, Farah MG. A Case of Infected Left Atrial Myxoma Presenting as ST-Elevation Myocardial Infarction (STEMI). Am J Case Rep 2019;20:1930–5. (In eng). DOI: 10.12659/ajcr.918192.
- Jayaprakash K, Cletus N, Jayakumar TK, et al. The masquerading myxoma – Presentation as acute STEMI. Journal of Indian College of Cardiology 2015;5(3):244–7. DOI: https://doi.org/10.1016/j.jicc.2015.01.002.
- Vondran M, Ghazy T, Andrási TB, et al. ST-Segment Elevation Myocardial Infarction and Right Atrial Myxoma. Thorac Cardiovasc Surg Rep 2022;11(1):e33–e37. (In eng). DOI: 10.1055/s-0042-1749211.
Article citation: Thiravetyan B, Soliman D, Ullah MQ, Sly Z. Silent culprit: Left atrial myxoma presenting as anterior STEMI. The Southwest Journal of Medicine. 2026;14(58):68–73
From: Department of Internal Medicine, Texas Tech University Health Sciences Center, Lubbock, TX (BT, DS, MQU, SZ)
Conflicts of interest: none
This work is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.