ABSTRACT
Background: Pericardial effusion presents with a wide clinical spectrum, ranging from incidental findings to life-threatening cardiac tamponade. Hemorrhagic pericardial effusions (HPE) are most associated with malignancy, autoimmune conditions, and tuberculosis; however, idiopathic cases are rarely reported and can pose diagnostic challenges.
Case: A 57-year-old female presented with progressive dyspnea, orthopnea, and dry cough. Echocardiography revealed a large pericardial effusion with tamponade physiology, prompting urgent pericardiocentesis with drainage of 800 mL of hemorrhagic fluid. Extensive infectious, malignant, and autoimmune workup was unrevealing, and pericardial biopsy showed no evidence of malignancy or infection. The patient was managed empirically for pericarditis with NSAIDs and colchicine. At a two-month follow-up, she demonstrated clinical and echocardiographic improvement without evidence of constrictive physiology.
Conclusion: Idiopathic hemorrhagic cardiac tamponade, although rare, must be considered when common etiologies are excluded. Timely recognition and intervention are critical to patient survival. Comprehensive evaluation and long-term follow-up remain essential to guide management and monitor for recurrence.
Keywords: Pericardial tamponade, hemorrhagic pericardial effusion.
KEY CLINICAL MESSAGE
Pericardial tamponade may present subtly without Beck’s triad. In hemorrhagic effusion, infection, autoimmune diseases, and malignancy should be ruled out. If the workup is negative, we suggest empirical treatment for acute pericarditis.
INTRODUCTION
Pericardial effusion encompasses a broad clinical spectrum, ranging from asymptomatic presentations to life-threatening cardiac tamponade. Its etiology is diverse, including infectious, malignant, metabolic, autoimmune, traumatic, and idiopathic causes. Hemorrhagic pericardial effusions are most frequently associated with trauma, aortic dissection, malignancy, iatrogenic injury, autoimmune diseases, and tuberculosis in endemic regions; however, rare idiopathic cases—often presumed to be of viral origin—have also been documented.
The classic Beck’s triad of cardiac tamponade—hypotension, elevated jugular venous pressure, and muffled heart sounds—is considered characteristic but may not be present in all cases. Presentation can initially be subtle, with symptoms and signs such as exertional dyspnea, which may progress to orthopnea, associated with fatigue, and tachycardia, ultimately culminating in circulatory collapse if not promptly recognized and managed.1
We present this case to raise awareness among physicians about a potentially life-threatening condition that may present with subtle or atypical symptoms. This case highlights the critical role of thorough physical examination and the utility of incorporating point-of-care ultrasound (POCUS) in timely diagnosis. Additionally, we summarize the recommended diagnostic workup for hemorrhagic pericardial tamponade to guide clinicians in identifying and managing this rare but serious clinical entity.
CASE
A 57-year-old female patient with medical history of well-controlled hypothyroidism and newly diagnosed hypertension reported a two-month history of progressively worsening shortness of breath and a dry cough, which became more pronounced with exertion and when lying flat. She denied any recent illness, or travel. Her primary care physician referred her for an outpatient echocardiogram, which revealed a large pericardial effusion with echocardiographic signs of tamponade (Figure 1, Videos 1, 2, 3). This critical finding prompted immediate notification of our cardiology team.
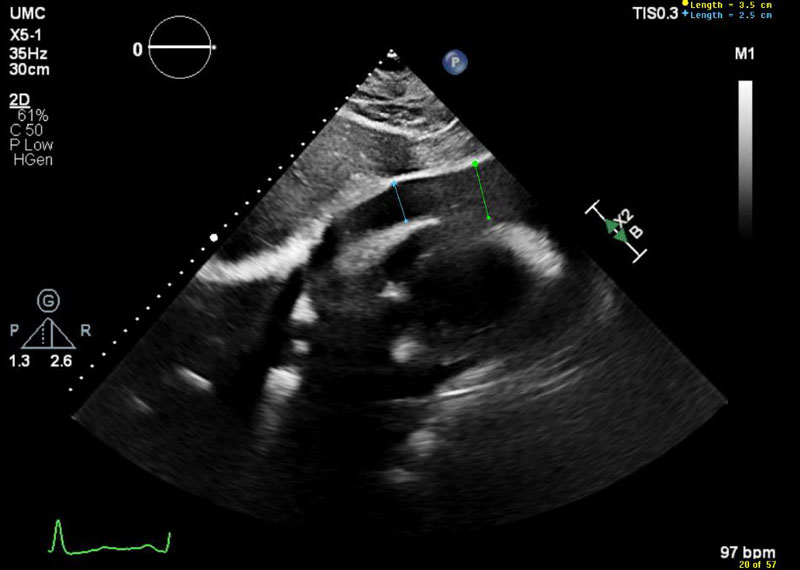
Figure 1 Subcostal view showing large pericardial effusion measuring up to 3.5 cm in diastole.
On examination, the patient exhibited sinus tachycardia with a resting heart rate of 112 bpm and a blood pressure of 148/112 mmHg. She had a positive pulsus paradoxus, a respiratory rate of 22 bpm, and auscultation revealed distant heart sounds, no pericardial rub, and bilateral clear lung sounds. The remainder of her physical exam was unremarkable. Given the severity of the findings, she was urgently transported to the cardiac catheterization lab for further evaluation and intervention.
The patient was placed under conscious sedation with continuous hemodynamic monitoring. Fluoroscopic and ultrasound-guided pericardiocentesis was performed, yielding 800 mL of brown-colored pericardial fluid, and a pigtail catheter was placed for continued drainage. Postoperatively, continuous hemodynamic monitoring demonstrated resolution of tachycardia and normalization of blood pressure. Over the following two days, the drainage output gradually decreased to less than 50 mL. A follow-up echocardiogram two days later demonstrated residual loculated pericardial effusion, pericardial thickening with septal bouncing, and Doppler findings suggested possible constrictive pericarditis (Figure 2).
Cardiothoracic surgery subsequently performed a pericardial window with biopsy, which revealed a thickened pericardial rind without evidence of malignancy or fungal infection. The patient was discharged on ibuprofen and colchicine empirically for treatment of pericarditis. At her two-month follow-up visit, she reported significant improvement in exercise tolerance and follow-up echo showed no evidence of constrictive pericarditis or pericardial effusion recurrence. The patient will continue follow-up at gradually increasing intervals to monitor for recurrence.
DISCUSSION
Hemorrhagic pericardial effusion (HPE) is a distinct form of pericardial effusion characterized by the accumulation of serosanguineous or frank blood within the pericardial cavity. Identifying the underlying etiology of HPE can be complex and multifactorial. Invasive cardiac procedures represent the most common cause, accounting for approximately 31% of hemopericardium cases. However, a wide range of other etiologies may contribute, including thoracic trauma, complications of ischemic heart disease, malignancy, infections, autoimmune diseases, use of anticoagulant therapy, chronic kidney disease, and idiopathic causes. 2 To our knowledge, no data is available on the prevalence of idiopathic hemorrhagic cardiac tamponade. A post-mortem analysis of 430,000 autopsies conducted between 1995 and 2004 identified 461 deaths attributable to cardiac tamponade secondary to hemopericardium, irrespective of the underlying etiology.1
The clinical presentation is influenced by the rate of pericardial effusion accumulation. In acute scenarios, relatively small effusions (<250 mL) may precipitate significant cardiac tamponade. Conversely, even large effusions (1,000–2,000 mL) may remain clinically silent in terms of tamponade if they develop gradually over time.3 The classic Beck’s triad—hypotension, elevated jugular venous pressure, and muffled heart sounds—is more readily recognized in acute clinical settings based on patient history and physical examination. However, numerous studies and clinical observations have demonstrated that this triad lacks both sensitivity and specificity for the diagnosis of cardiac tamponade. A retrospective study of emergency department admissions conducted in 2017 reported a sensitivity of 0% (range: 0–19.4%) for the triad in identifying tamponade.4 Diagnostic challenges are particularly pronounced in subacute presentations of pericardial effusion and cardiac tamponade. In our case, the patient presented with nonspecific symptoms, including fatigue, exertional dyspnea, orthopnea, and dry cough—clinical features that may also be attributed to other less acute conditions such as heart failure or pulmonary hypertension. On physical examination, resting tachycardia was the most notable finding, while other classical signs such as muffled heart sounds and elevated jugular venous pressure were potentially obscured by the patient’s body habitus. This underscores the importance of maintaining a high index of suspicion for life-threatening conditions such as cardiac tamponade, even in the presence of subtle or atypical clinical findings. Moreover, the integration of point-of-care ultrasound (POCUS) into routine clinical practice has proven invaluable in the timely identification of critical cardiac pathology.
In the 2015 ESC guidelines on pericardial diseases, transthoracic echocardiography is recommended as the single most important diagnostic tool for the detection of cardiac tamponade.5 Major echocardiographic findings include swinging, pendular motion of the heart within the pericardial fluid, collapse of the right atrium during late diastole, collapse of the right ventricle during early diastole, compression of the left atrium and, rarely, the left ventricle. Echo also is important for the diagnosis of effusive constrictive pericarditis, and for the follow-up after treatment to identify pericardial effusion recurrence or development of pulmonary hypertension after treatment. Invasive right heart catheterization might be indicated at the time of pericardiocentesis if effusive constrictive pericarditis is suspected. Electrocardiography (EKG) typically shows low QRS voltage and electrical alternans. Chest radiograph has water-bottle shaped appearance of the heart with cardiomegaly. Pericardial fluid analysis is mandatory for assessment of underlying etiology of pericardial effusion. In cases of hemorrhagic pericardial effusion without a history of trauma or anticoagulant use, a comprehensive evaluation to exclude malignancy, autoimmune disorders, and tuberculosis should be undertaken before classifying the effusion as idiopathic.1
In cases of acute hemorrhagic pericardial tamponade associated with anemia, hemodynamic instability, or a history of trauma, Type A aortic dissection should be ruled out prior to performing pericardiocentesis. This is best accomplished with computed tomography angiography (CTA) of the chest or transesophageal echocardiography (TEE).
In suspected malignancy-associated effusions, pericardial fluid analysis should include cytological examination and assessment of tumor markers such as carcinoembryonic antigen (CEA), CA 19-9, CA 72-4, CA 15-3, CA 125, CYFRA 21-1, alpha-fetoprotein (AFP), and adenosine deaminase (ADA). Pericardial biopsy, along with a thorough oncologic evaluation—including chest computed tomography (CT), colonoscopy, mammography, and breast ultrasonography—is recommended to establish a diagnosis.
In cases of infectious etiology, diagnostic evaluation should include Gram staining, acid-fast bacillus (AFB) staining, and cultures for bacterial (including mycobacterial) and fungal organisms. Based on the clinical history, additional tests may be warranted, such as interferon-gamma release assays or lysozyme levels, HIV serology, and PCR assays for viruses known to cause pericarditis, including Influenza A, respiratory syncytial virus (RSV), dengue virus, chikungunya virus, coronaviruses, parvovirus B19, Epstein–Barr virus (EBV), and human herpesvirus 6 (HHV-6).
For autoimmune-related pericardial effusions, initial serologic screening should include antinuclear antibody (ANA), anti–double-stranded DNA (anti-dsDNA), anti-Smith antibodies, complement levels (C3 and C4), rheumatoid factor (RF), and anti–cyclic citrullinated peptide (anti-CCP) antibodies. In cases where initial testing is inconclusive, extended autoimmune panels, including anti–Sjögren’s syndrome-related antigen A (anti-SSA), anti–Sjögren’s syndrome-related antigen B (anti-SSB), anti-CREST antibodies (associated with calcinosis, Raynaud phenomenon, esophageal dysmotility, sclerodactyly, and telangiectasia), anti-Scl-70, anti–ribonucleoprotein (anti-RNP), anti–PL-7, anti–PL-12, perinuclear anti-neutrophil cytoplasmic antibodies (p-ANCA), cytoplasmic ANCA (c-ANCA), anti–hepatitis C virus (anti-HCV) antibodies, and myositis-specific antibody panels.2
If left untreated, cardiac tamponade is uniformly fatal. Prompt intervention through early pericardial drainage—whether performed via percutaneous ultrasound- or fluoroscopy-guided pericardiocentesis, or through the creation of a surgical pericardial window—is associated with significantly improved clinical outcomes. Ongoing follow-up is essential to monitor for recurrence of pericardial effusion. Further management should be guided by the underlying etiology.1
CONCLUSION
Hemorrhagic pericardial effusion, though often secondary to identifiable causes, may rarely be present as idiopathic and poses a significant diagnostic challenge. Early recognition and prompt intervention are critical to preventing the progression to fatal cardiac tamponade. Long-term management should be guided by the underlying etiology, with close follow-up to monitor for recurrence.
VIDEOS
Video 1. Parasternal short-axis echocardiographic view demonstrating a large circumferential pericardial effusion.
Video 2. Apical four-chamber echocardiographic view demonstrating a large pericardial effusion with right ventricular (RV) diastolic collapse, consistent with tamponade physiology.
Video 3. Parasternal long-axis echocardiographic view demonstrating a large pericardial effusion with right ventricular (RV) collapse, suggestive of cardiac tamponade.
REFERENCES
- Adler Y, Ristic AD, Imazo M, et al. Cardiac tamponade. Nature Reviews Disease Primers. 2023;9(1):36.
- Borkowski P, Borkowska N, Nazarenko N, et al. Hemopericardium: a comprehensive clinical review of etiology and diagnosis. Cureus. 2024;16(1).
- Jeong T-D, Jang S, Park C-J, et al. Prognostic relevance of pericardial effusion in patients with malignant diseases. The Korean Journal of Hematology. 2012;47(3):237–8.
- Stolz L, Valenzuela J, Situ-LaCasse E, et al. Clinical and historical features of emergency department patients with pericardial effusions. World Journal of Emergency Medicine. 2017;8(1):29.
- Adler Y, Charron P. 2015 ESC Guidelines for the diagnosis and management of pericardial diseases. Polish Heart Journal (Kardiologia Polska). 2015;73(11):1028–91.
Article citation: Soliman D, Ashour F, Castillo-Rodriguez C, Puchongmart C, Abdelmalek J. Hemorrhagic cardiac tamponade presenting as isolated exertional dyspnea in an otherwise healthy individual: A diagnostic challenge. The Southwest Journal of Medicine. 2026;14(59):88–93
From: Texas Tech University Health Sciences Center, Lubbock, TX (DS, FA, CC-R, CP, JA)
Conflicts of interest: none
This work is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.