ABSTRACT
A man in his early 40’s with Klippel-Trenaunay-Weber syndrome (KTWS) was referred to the surgical team for treatment of massive upper gastrointestinal bleeding. This syndrome is a rare congenital condition that leads to abnormal development of blood vessels, bones, and soft tissues. Although venous malformations are common in KTWS, gastrointestinal involvement is rare but can cause life-threatening bleeding. In this case, a series of diagnostic tests, including endoscopic and angiographic examinations, failed to identify the etiology of the bleeding. Surgical intervention was required after conservative and interventional radiologic therapies proved unsuccessful. A Dieulafoy’s lesion was identified in the proximal stomach and later confirmed histologically. The patient required a multidisciplinary care involving gastroenterology, interventional radiology, and general surgery for successful management.
The pathogenesis of Dieulafoy’s lesion is not well understood, and interventions vary from endoscopic to surgical management. The treatment of Dieulafoy’s lesion is not well- described in patients with KTWS, especially in cases in which endoscopic treatments have failed.
Keywords: Klippel-Trenaunay Syndrome; Dieulafoy’s lesion; gastrointestinal bleeding; radiology, interventional; endoscopy; surgical treatment
INTRODUCTION
Klippel-Trenaunay-Weber syndrome (KTWS) is caused by a somatic mutation in the PIK3CA gene, resulting in abnormal development of blood vessels, bones, and soft tissues. This manifests as varicose veins, port-wine stains, and musculoskeletal overgrowth, often associated with bleeding disorders.1 Given its rare prevalence of 2–5 cases per 100,000 live births, diagnosis is usually based on clinical evaluation with no established guidelines.2 Bleeding in KTWS is due to venous malformations, and although gastrointestinal involvement is rare, it can lead to life-threatening hemorrhage. One such manifestation is a Dieulafoy’s lesion, a rare vascular malformation characterized by abnormal dilation of a submucosal artery, causing a small but significant bleeding vessel without ulcers or erosions. In KTWS, this may result from the vascular abnormalities caused by the PIK3CA mutation, but other theories suggest mucosal thinning or arterial pulsation leading to ischemia.3 Dieulafoy’s lesions account for 1–2% of gastrointestinal bleeds and 6.5% of upper gastrointestinal (GI) hemorrhages.4,5 This case presents the first documented surgical intervention for a Dieulafoy’s lesion in a KTWS patient, highlighting both the diagnostic challenges of Dieulafoy’s lesions and the added complexity of managing KTWS, particularly the increased thrombotic risk due to vascular abnormalities.
CASE
A man in his early 40s with a history of KTWS, hypertension, hyperlipidemia, and major depressive disorder presented to the emergency department after two episodes of melena and hematemesis, accompanied by chills, diaphoresis, and near-syncope. Two weeks prior, he had a motor vehicle accident and was prescribed high-dose ibuprofen (1000 mg daily) for muscle soreness. He had also been non-compliant with low-dose aspirin therapy prescribed for KTWS.
On examination, the patient was afebrile and normotensive. Skin examination revealed port-wine stains on the lower extremities, consistent with his KTWS. Abdominal examination was unremarkable. Laboratory tests showed a normal CBC and basic metabolic panel, except for elevated BUN (30 mg/dL). A thromboelastogram revealed normal fibrinogen function and fibrinolysis and hypercoagulable platelet function. Imaging, including upright chest x-ray and computed tomography (CT) of the abdomen and pelvis, did not identify any acute GI bleeding.
Initial suspicion was for NSAID-induced gastritis or duodenal ulcer given his ibuprofen use. The following day, endoscopy revealed grade 1 esophageal varices, two non-bleeding ulcers at the gastroesophageal junction (Figure 1A), hemorrhagic gastritis, and duodenal ulcers in D1 and D2 (Figure 1B). Five clips were placed to stop the bleeding, and the patient was managed with proton pump inhibitors and sucralfate, with follow-up planned in six weeks.
Despite initial control, the patient experienced recurrent hematemesis with his hemoglobin decreasing from 13.4 g/dL to 8.0 g/dL. Epinephrine was administered locally, but no active bleeding was identified. With worsening hemodynamic instability, interventional radiology was consulted, and a CT with angiography was performed. The angiogram showed no active GI bleed but did reveal a large intragastric blood clot (12 cm). The patient was transferred to the ICU after a 30-hour bleeding-free interval, and interventional radiology performed an empiric embolization of the gastroduodenal artery using 500–700-micron particles. However, this failed to achieve adequate control of the bleeding (Figure 2).
With hemoglobin levels continuing to drop (7.2 g/dL), a massive transfusion protocol was activated, and the patient was taken emergently to the operating room. During exploratory laparotomy, the stomach was distended, and 2.5 liters of clotted blood were evacuated. A bleeding Dieulafoy’s lesion was identified and oversewn, achieving definitive hemostasis. The pathology report confirmed the diagnosis. Postoperatively, the patient’s hemodynamic status improved, and he was transferred to the surgical ICU.
The patient’s recovery was complicated by deep vein thromboses in both the upper and lower extremities. Vascular surgery was consulted, and a CT aortogram with bilateral lower extremity runoff revealed no signs of arterial occlusion or stenosis (Figure 3). By postoperative day 5, his hemoglobin remained stable, and an upper GI series showed no leaks, allowing his diet to be advanced. By day 15, the patient was ambulating independently and was discharged on apixaban with follow-up scheduled. He was seen in the outpatient clinic without any additional complications.
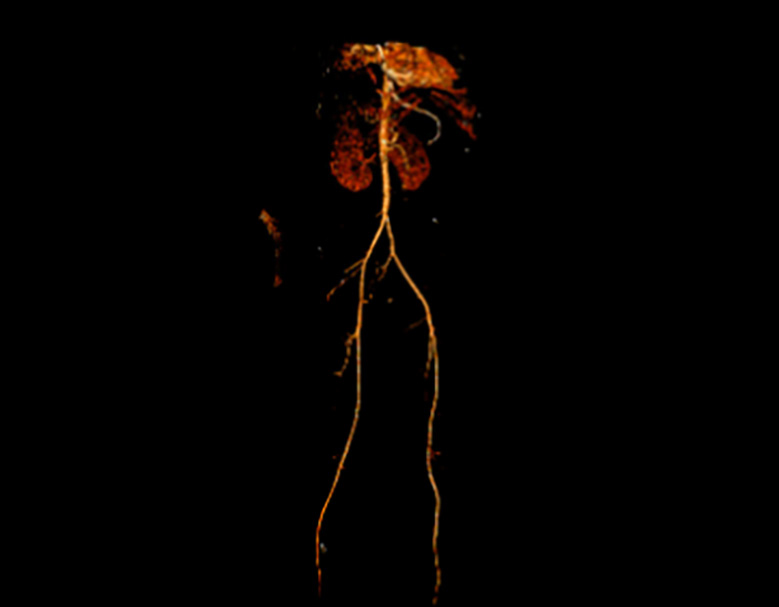
Figure 3. A CT Aortogram with bilateral lower extremity runoff via the posterior tibial artery shows no evidence of thrombus or embolus.
DISCUSSION
In this case, the first-line treatment for Dieulafoy’s lesions, endoscopic therapy, was ineffective, necessitating surgical intervention. Despite initial attempts with epinephrine injection and mechanical clipping, as described in previous reports, these measures were insufficient to control the bleeding. The complexity of managing GI bleeding in KTWS patients, particularly in the presence of vascular malformations, highlights the need for a multidisciplinary approach.
Endoscopic management, as described by Jeon and Kim (2015), typically achieves success rates of 75% to 100% using epinephrine and mechanical techniques.3,6 However, these methods were unsuccessful in our case, necessitating further intervention. Dadhania et al. (2021) emphasized the effectiveness of CT angiography and interventional radiology in treating Dieulafoy’s lesions after endoscopic failure, while Batouli et al. (2015) highlighted its value in diagnosing difficult bleeds.7,8 In our patient, however, interventional radiology-guided embolization of the gastroduodenal artery, as suggested by Dadhania et al., failed to fully control the bleeding, underscoring the risk of ischemia when dealing with multiple collateral vessels.7 Surgical intervention, while less common, remains a critical option in refractory cases. As noted by Joarder et al. (2014), surgery is required in about 5% of patients when endoscopic and angiographic methods prove insufficient.9 In our case, the use of open gastrotomy and oversewing was consistent with the approach outlined by Alva et al. (2006), in which wedge resection and direct suturing have proven effective in achieving hemostasis.10 Furthermore, laparoscopic surgery, while beneficial in many cases, was not feasible for our patient due to the extensive bleeding and difficult location of the lesion, echoing challenges noted by Alva et al. and Joarder et al.9,11
The association between KTWS and GI bleeding, including vascular malformations like Dieulafoy’s lesion, remains underexplored. Studies, such as those by Samo et al. (2013), have underscored the need for further investigation into the prevalence and pathophysiology of GI involvement in KTWS.12 Given the significant risk of life-threatening bleeding in these patients, future research should aim to better understand how KTWS predisposes individuals to Dieulafoy’s lesion and other arteriovenous malformations.
This report highlights a rare cause of upper GI bleed, but the management described has limitations. The multidisciplinary approach in this case was successful but may not be feasible in all healthcare settings due to resource limitations and access to specialized surgical teams. As with many rare disease case reports, broader studies are necessary to establish more concrete treatment guidelines.
REFERENCES
- Bertino F, Braithwaite KA, Hawkins CM, et al. Congenital limb overgrowth syndromes associated with vascular anomalies. RadioGraphics 2019;39(2):491–515.
- Asghar F, Aqeel R, Farooque U, Haq A, Taimur M. Presentation and management of Klippel-Trenaunay syndrome: a review of available data. Cureus 2020;12(5):e8023.
- Jeon HK, Kim GH. Endoscopic management of Dieulafoy’s lesion. Clin Endosc 2015;48(2):112–20.
- Marangoni G, Cresswell AB, Faraj W, et al. An uncommon cause of life-threatening gastrointestinal bleeding: 2 synchronous Dieulafoy lesions. J Pediatric Surgery 2009;44(2):441–3.
- Mansfield G, Sabharwal R, Bansi DS, et al. Dieulafoy lesions: rare but often overdiagnosed? Observations based upon a case of small bowel haemorrhage and a critical review of the literature. Clin Radiol 2012;67(1):78–80.
- Stark ME, Gostout CJ, Balm RK. Clinical features and endoscopic management of Dieulafoy’s disease. Gastrointestinal Endoscopy 1992;38(5):545–50.
- Dadhania D, Valakkada J, Ayyappan A, et al. Role of imaging and endovascular radiology in endoscopically missed Dieulafoy’s lesion of stomach – A case report with review. BJR Case Rep 2022;7(6):20210117.
- Batouli A, Kazemi A, Hartman MS, et al. Dieulafoy lesion: CT diagnosis of this lesser-known cause of gastrointestinal bleeding. Clinical Radiology 2015;70(6):661–6.
- Joarder AI, Faruque MS, Nur-E-Elahi M, et al. Dieulafoy’s lesion: an overview. Mymensingh Med J 2014;23(1):186–94.
- Baxter M, Aly EH. Dieulafoy’s lesion: current trends in diagnosis and management. Ann R Coll Surg Engl 2010;92(7):548–54.
- Alva S, Abir F, Tran DD. Laparoscopic gastric wedge resection for Dieulafoy’s disease following preoperative endoscopic localization with India ink and endoscopic clips. JSLS 2006;10(2):244–6.
- Samo S, Sherid M, Husein H, et al. Klippel-Trenaunay syndrome causing life-threatening GI bleeding: a case report and review of the literature. Case Rep Gastrointest Med 2013; 2013:813653.
Article citation: Karim S, Pert L, Khan A, Santos A. Surgical management and multidisciplinary care of Dieulafoy’s lesion in Klippel-Trenaunay-Weber Syndrome. The Southwest Respiratory and Critical Care Chronicles 2025;13(54):27–30
From: Texas Tech University Health Science Center School of Medicine (SK, LP, AS) Lubbock, TX; Medical College of Georgia (AK), Augusta, GA
Submitted: 9/17/2024
Accepted: 12/11/2024
Conflicts of interest: none
This work is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.